Key takeaways
- ●BPC-157 acts through multiple mechanisms including VEGF-driven angiogenesis, nitric oxide modulation, and FAK-paxillin cell migration signaling.
- ●It has unusual gastric stability that allows both oral and injectable research routes — rare for a peptide.
- ●Research spans tendon, muscle, GI, bone, nerve, and vascular tissue types, though nearly all data is preclinical.
- ●Sourcing quality depends on HPLC purity above 98%, batch-specific COAs, and cold-chain storage discipline.
BPC-157 is a short synthetic peptide — 15 amino acids — derived from a protective compound found in human gastric juice, and it is one of the most widely studied research peptides for tissue-repair models across animal research.
The unusual thing about it is stability. Most peptides break down quickly in stomach acid. BPC-157 survives it, which is why researchers have been able to investigate it through both injection and oral routes in animal models.
Origin and structure
BPC stands for body protection compound, a larger natural protein found in human gastric juice that appears to protect the stomach lining from its own acid. Researchers in the 1990s identified a 15-amino-acid fragment of that larger protein and synthesized it as a stand-alone molecule. That fragment is BPC-157.
The sequence is Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val. Molecular weight roughly 1,419 Da. CAS number 137525-51-0. It is a partial sequence of the parent protein, not the full protein — which is why the naming includes the number 157.
How it works
Research to date points to several mechanisms acting together rather than a single dominant pathway.
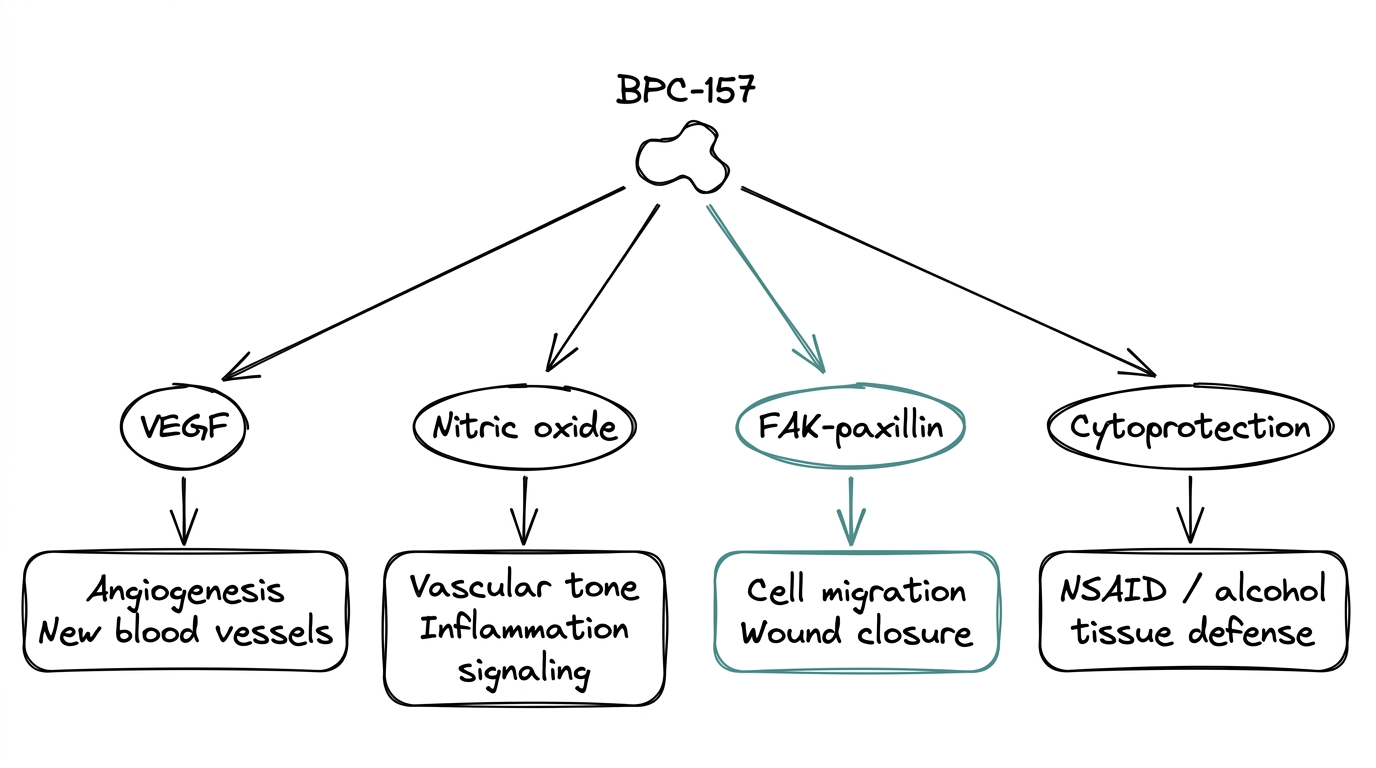
Angiogenesis and the VEGF pathway
BPC-157 upregulates vascular endothelial growth factor signaling in injury models. VEGF is the signal that tells the body to build new blood vessels, and fresh vasculature is what delivers oxygen and nutrients to repairing tissue. The angiogenesis effect is the most consistently replicated mechanism in the BPC-157 literature.
Nitric oxide modulation
Nitric oxide, or NO, is a small signaling molecule involved in blood-vessel tone, inflammation, and cell signaling. BPC-157 appears to modulate the NO system rather than simply boost or block it — protective in NO-depleted states and regulatory when NO is elevated. That dual behavior is unusual and is what makes the mechanism interesting to researchers studying vascular and inflammatory pathways.
FAK-paxillin and cell migration
Focal adhesion kinase (FAK) and its partner protein paxillin are part of the machinery cells use to pull themselves across a surface. Wound closure requires cells to migrate into the damaged area. Studies show BPC-157 activates FAK-paxillin signaling, which is the mechanistic link between BPC-157 and measurable wound-closure outcomes in animal models.
Growth-factor regulation
BPC-157 appears to influence the availability and signaling of repair-related growth factors including EGF (epidermal growth factor) and FGF (fibroblast growth factor). These are the signals that coordinate the phases of tissue repair. The effect is regulatory rather than replacement — BPC-157 is not a growth factor itself, but it changes how the body uses its own.
Cytoprotection
In animal models, BPC-157 shows protective effects against damage from NSAIDs, alcohol, and various toxin exposures. The effect is strongest in gastrointestinal tissue, which tracks with the compound's origin in gastric juice — but cytoprotective effects extend to liver, kidney, and nerve tissue in different models.
Tissue types researchers have studied
BPC-157 has been investigated across an unusually broad range of tissue types. That breadth is part of why it is one of the most-ordered research peptides.
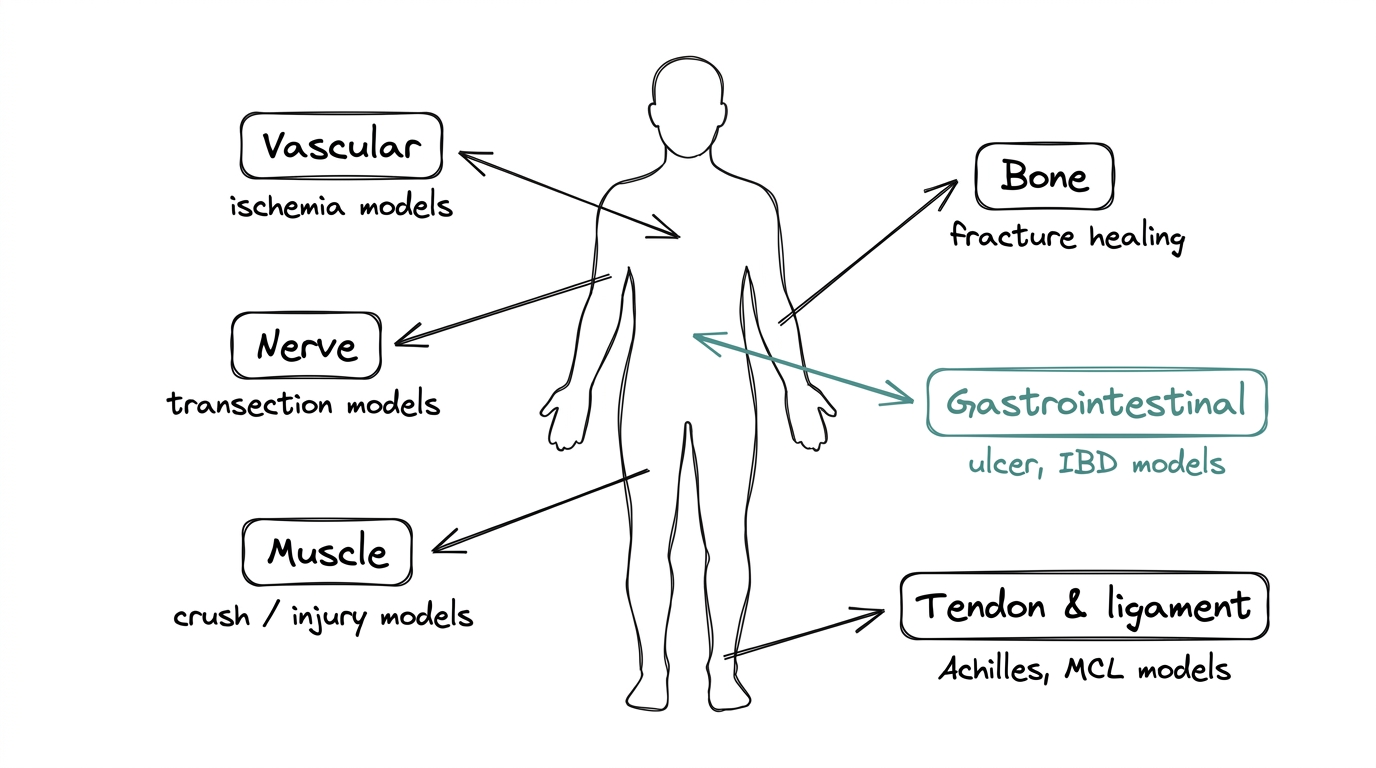
The main research areas, with typical model types:
Tendon and ligament
Achilles tendon transection and MCL injury models in rodents. This is the most well-known cluster of studies, cited heavily by recovery-research communities.
Muscle
Muscle-crush and muscle-injury models showing faster recovery of contractile function in treated animals.
Gastrointestinal tract
Ulcer and inflammatory-bowel models, where the gastric-origin hypothesis first led researchers to study BPC-157.
Bone
Fracture-healing models showing accelerated mineralization and callus formation.
Nerve and vascular tissue
Nerve-transection and ischemia models looking at whether the angiogenesis and FAK-paxillin effects translate to neural regeneration.
Across all of these, the literature is primarily rodent studies. The gap between animal results and human clinical data is a genuine discussion point in the field and worth being honest about.
Oral versus injectable in research
Most peptides cannot be dosed orally because stomach acid degrades them before they can be absorbed.
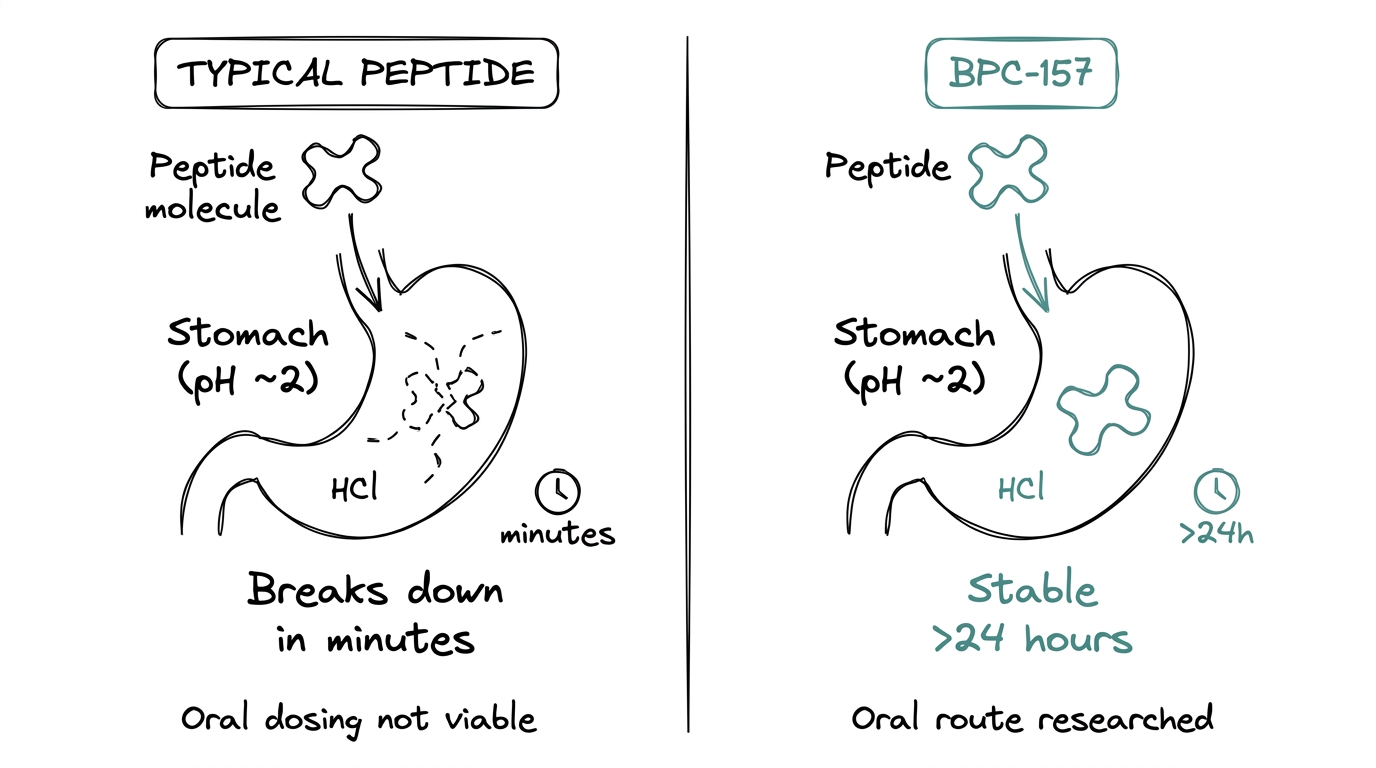
BPC-157 is different. The same gastric-juice origin that gave it its name also gave it unusual acid stability — research shows it survives more than 24 hours in stomach-pH conditions. Oral bioavailability in animal studies ranges roughly 10 to 30 percent, which is low compared to injection but high enough for the oral route to be researched seriously. Both injectable and oral protocols appear in the published literature.
This is the piece of BPC-157 pharmacology that distinguishes it from almost every other research peptide. Oral routes are rarely an option elsewhere.
What researchers look for in sourcing
For research use, three things determine whether a BPC-157 supply is fit for purpose.
Purity
HPLC purity above 98 percent is the research-grade floor. Lower purity means more unknown material in every dose, which means more variables in every experiment.
Certificate of Analysis per batch
A batch-specific COA showing peptide content, HPLC trace, and mass spectrometry identification is what separates a real research supply from repackaged material. A product-line COA (one certificate covering multiple batches) is not the same thing.
Storage discipline in transit
Lyophilized BPC-157 is stable at ambient temperature for short windows but should be stored at −20 °C long-term and 2–8 °C once reconstituted. Suppliers who ship without cold-chain awareness do not consistently deliver intact peptide.
For a deeper walk-through of what a real COA shows and the red flags that usually mean repackaged material, the peptide COA quality guide covers the verification steps in detail.
Limits and tradeoffs
The literature on BPC-157 is almost entirely preclinical. Human trials are limited in number and in scope, and translating rodent-dosing data to human-equivalent doses is where most claims in the wider BPC-157 discussion go wrong. Animal data is animal data.
BPC-157 is not an FDA-approved therapy and is not a dietary supplement under current US regulation. It is supplied for research use only. The World Anti-Doping Agency lists it as prohibited for competitive athletes, which is worth knowing if the research context involves sports-medicine work.
The recoverability of specific findings across labs is another real limit. Angiogenesis and FAK-paxillin effects replicate consistently. Some of the broader effects — particularly around neural regeneration — are more lab-dependent and worth reading the original studies to evaluate.
Where to go from here
Reference compound for tissue-repair research
BPC-157 is the reference compound for tissue-repair research questions. The breadth of tissue types studied, the mechanism coverage across angiogenesis and cell migration, and the unusual oral bioavailability are what make it a first-line candidate.
For researchers comparing BPC-157 to TB-500 — the other commonly cited tissue-repair peptide — the BPC-157 vs TB-500 comparison covers the mechanism differences and where each one fits.
For the quality-verification side, the peptide COA quality guide walks through what a real Certificate of Analysis looks like and the red flags that signal repackaged material.
Frequently asked questions
What is BPC-157?
BPC-157 is a synthetic 15-amino-acid peptide derived from a protective protein found in human gastric juice. It is studied in animal models for tissue-repair signaling across multiple tissue types.
How does BPC-157 work mechanistically?
BPC-157 acts through several pathways including VEGF-driven angiogenesis (new blood vessel formation), nitric oxide modulation, FAK-paxillin cell migration signaling, growth-factor regulation, and cytoprotection against toxin exposure.
Can BPC-157 be taken orally?
Unlike most peptides, BPC-157 has unusual acid stability and survives more than 24 hours in stomach-pH conditions. Both oral and injectable routes appear in the published research literature.
What tissue types has BPC-157 been studied in?
Research covers tendon and ligament, muscle, gastrointestinal tract, bone, nerve, and vascular tissue — primarily in rodent models.
Is BPC-157 FDA approved?
No. BPC-157 is not an FDA-approved therapy and is not a dietary supplement. It is supplied for research use only. WADA also lists it as prohibited for competitive athletes.
What purity should research-grade BPC-157 have?
HPLC purity above 98 percent is the research-grade floor. Each vial should ship with a batch-specific Certificate of Analysis showing peptide content, HPLC trace, and mass spectrometry identification.
Next step
Source research-grade BPC-157
Batch-specific COAs, HPLC purity above 98%, and cold-chain shipping for every order.