Key takeaways
- ●A COA is batch-specific — a document covering an entire product line is marketing material, not a real certificate.
- ●HPLC purity and peptide content are different measurements; content (net peptide) is often more useful for dose accuracy.
- ●The six-point verification checklist covers lot number, HPLC purity with chromatogram, mass spec, peptide content, water content, and analyst signature.
- ●Four red flags account for most bad COAs: template reuse across lots, purity without chromatogram, in-house only testing, and gated access.
A Certificate of Analysis, or COA, is the batch-level test report a research peptide should ship with. Reading one properly is the single cleanest way to verify what is actually in the vial.
This guide walks through every field on a real COA, explains the analytical methods behind the numbers, and gives researchers a six-point checklist for evaluating any vendor's documentation.
What a COA is
A COA is a batch-level document, not a product-level document. That distinction is the most important thing to understand before reading one.
Two vials of the same peptide from the same vendor can belong to different manufacturing lots with different purity profiles, different water content, and different counter-ion ratios. A real COA is generated from tests performed on the specific lot that produced the specific vial being sold. A document that covers an entire product line is not a COA in the meaningful sense — it is marketing material with the word "Certificate" on it.
A legitimate research supplier produces a COA per batch, with a batch number that matches the label on the vial. The analytical tests behind it are usually run by a third-party analytical lab, though in-house testing also appears in the market and is not automatically disqualifying.
The fields on a COA, line by line
Every field on a well-prepared COA answers a specific question about the material. Here is what each one is asking and how to read the answer.
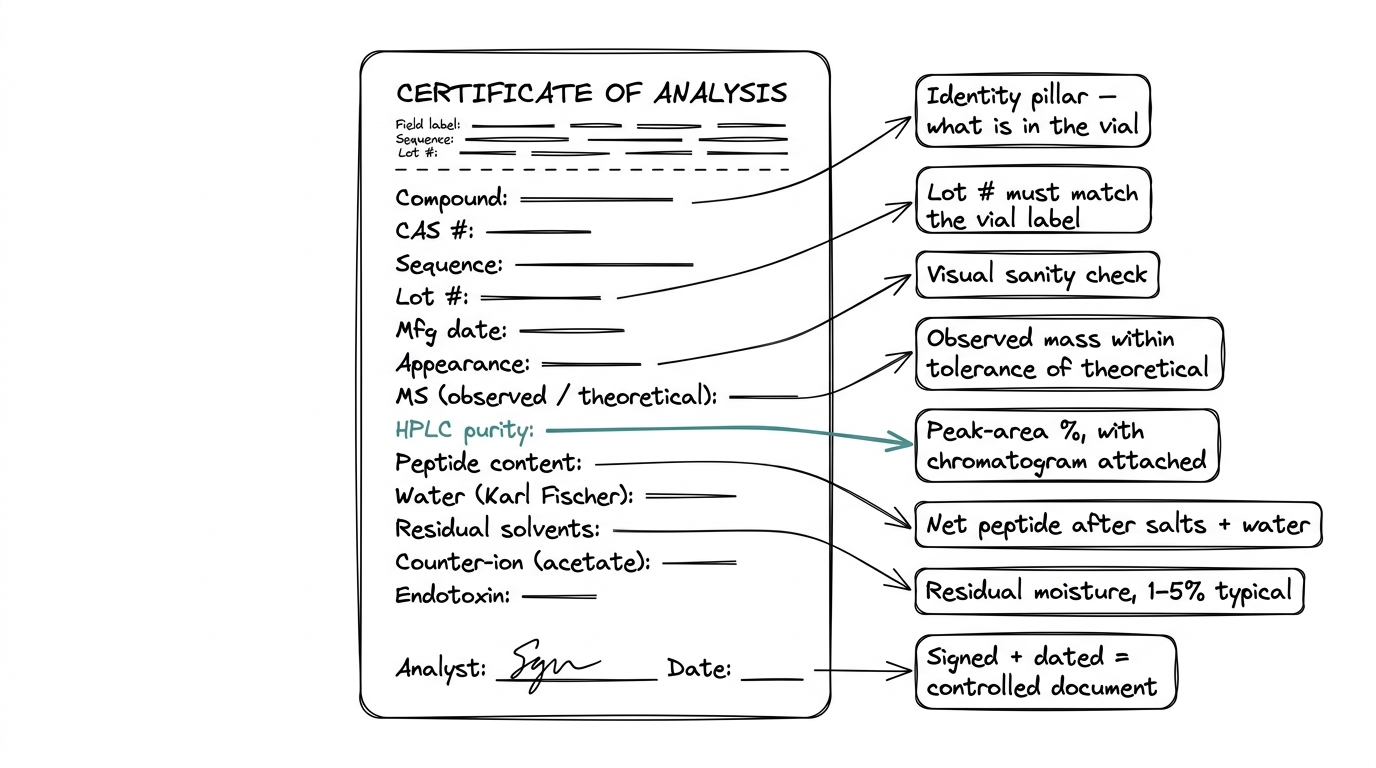
Product identification
Compound name, CAS number, sequence, molecular formula, molecular weight. This is the identity pillar. The COA must name exactly what the vial contains. A missing CAS number or sequence is the first sign that the document is not rigorous.
Lot number and manufacturing date
The batch-specific traceability back to a production run. A COA without a lot number cannot be tied to the vial in hand, which defeats the purpose of having a COA at all.
Appearance
Physical description of the lyophilized powder — usually "white to off-white lyophilized powder." A quick visual check against this description when the vial arrives is the first sanity check before anything else.
Identity confirmation by mass spectrometry
Mass spectrometry, or MS, measures the exact molecular mass of the compound in the vial. The COA reports the observed mass, which should match the theoretical mass for the peptide within a narrow tolerance. For BPC-157, that is roughly 1,419 Da. A large discrepancy between observed and theoretical mass means the vial does not contain what the label says it contains.
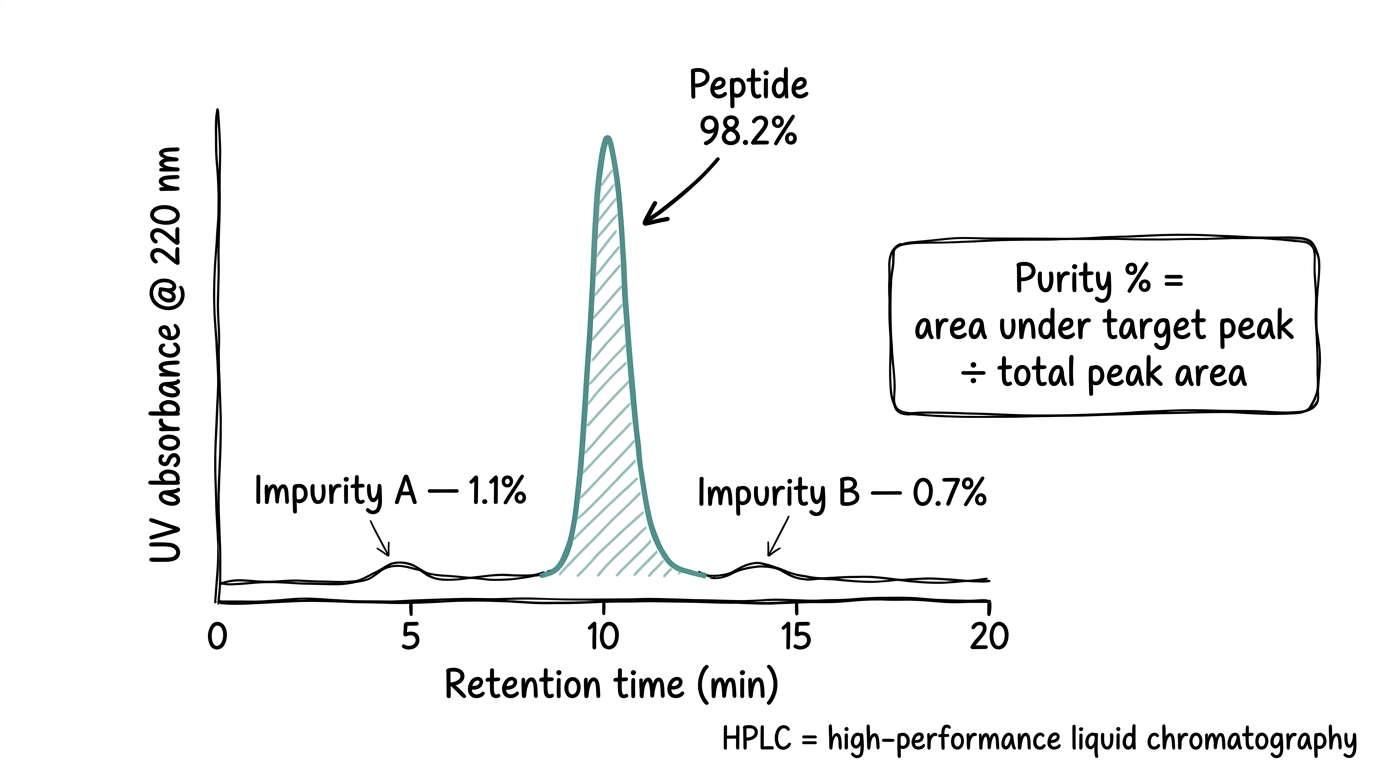
Purity by HPLC
High-performance liquid chromatography, or HPLC, separates the sample into its individual components and measures how much of the total is the target peptide versus impurities. The purity percentage on a COA is the area under the main peak expressed as a percentage of all peak area in the chromatogram.
Research-grade peptides should be greater than 95% pure. High-grade is 98% or above. A COA that reports purity without an attached chromatogram gives the number without the evidence — the chromatogram is how the number is actually substantiated.
Peptide content
Peptide content and peptide purity are different measurements, and the distinction catches a lot of buyers off guard. Purity is how clean the peptide fraction is. Content — sometimes called net peptide — is how much of the vial's total mass is actually peptide after water, counter-ions, and salts are accounted for.
A vial labeled 10 mg with 80% peptide content delivers roughly 8 mg of actual peptide. For research work where dose accuracy matters, peptide content is often the more useful number.
Water content by Karl Fischer
Karl Fischer titration measures the residual moisture in the lyophilized powder. Typical range is 1 to 5 percent. Too much water suggests incomplete lyophilization, which affects both potency and shelf life.
Residual solvents
Leftover manufacturing solvents — typically acetonitrile, trifluoroacetic acid, or methanol. These should sit within ICH Q3C guideline limits. Most COAs report these as trace amounts or below the limit of detection.
Counter-ion or acetate content
Most research peptides are supplied as acetate salts. The counter-ion takes up mass in the vial, which is why peptide content sits below 100 percent of the stated weight. The counter-ion figure feeds directly into any peptide-content calculation.
Endotoxin, when applicable
For material intended for certain animal-model work, endotoxin levels are measured by the Limulus Amebocyte Lysate (LAL) test and reported in endotoxin units per milligram. Not every COA includes it — endotoxin testing is selective, driven by the research application.
Analyst signature and date
A COA that is not signed off by a named analytical chemist and dated is an uncontrolled document. That field is easy to overlook, and its absence is diagnostic.
The analytical methods behind the numbers
Four methods do most of the work on a peptide COA. Each answers a specific question.
| Method | Question it answers | Technique |
|---|---|---|
| HPLC | How pure is the peptide fraction? | UV detection at 220 nm, purity is peak-area based |
| Mass spectrometry | Is it actually the peptide I ordered? | ESI-MS, mass accurate to within a few daltons |
| Karl Fischer titration | How much water is in the powder? | Direct chemical reaction specific to water |
| LAL | Is the sample free of bacterial endotoxin? | Protects downstream animal-model work from inflammatory artifacts |
Some higher-grade COAs also include amino acid analysis or NMR data. Both are optional and signal a more rigorous analytical program.
A six-point verification checklist
Most COA evaluations collapse to six questions. If every answer is yes, the document is reliable. If any answer is no, ask for a better document or find a different supplier.

- ●Does the COA name a specific lot number that matches the vial label?
- ●Is HPLC purity reported at 95 percent or higher, with a chromatogram or trace attached?
- ●Is the mass-spec observed mass within acceptable range of the theoretical mass?
- ●Is peptide content (net peptide) stated as a separate number from purity?
- ●Is water content reported?
- ●Is the COA signed by an analytical chemist and dated?
The checklist is short on purpose. It is designed to be applied in under a minute per document.
Red flags in vendor COAs
Four patterns account for almost every bad COA in the market.
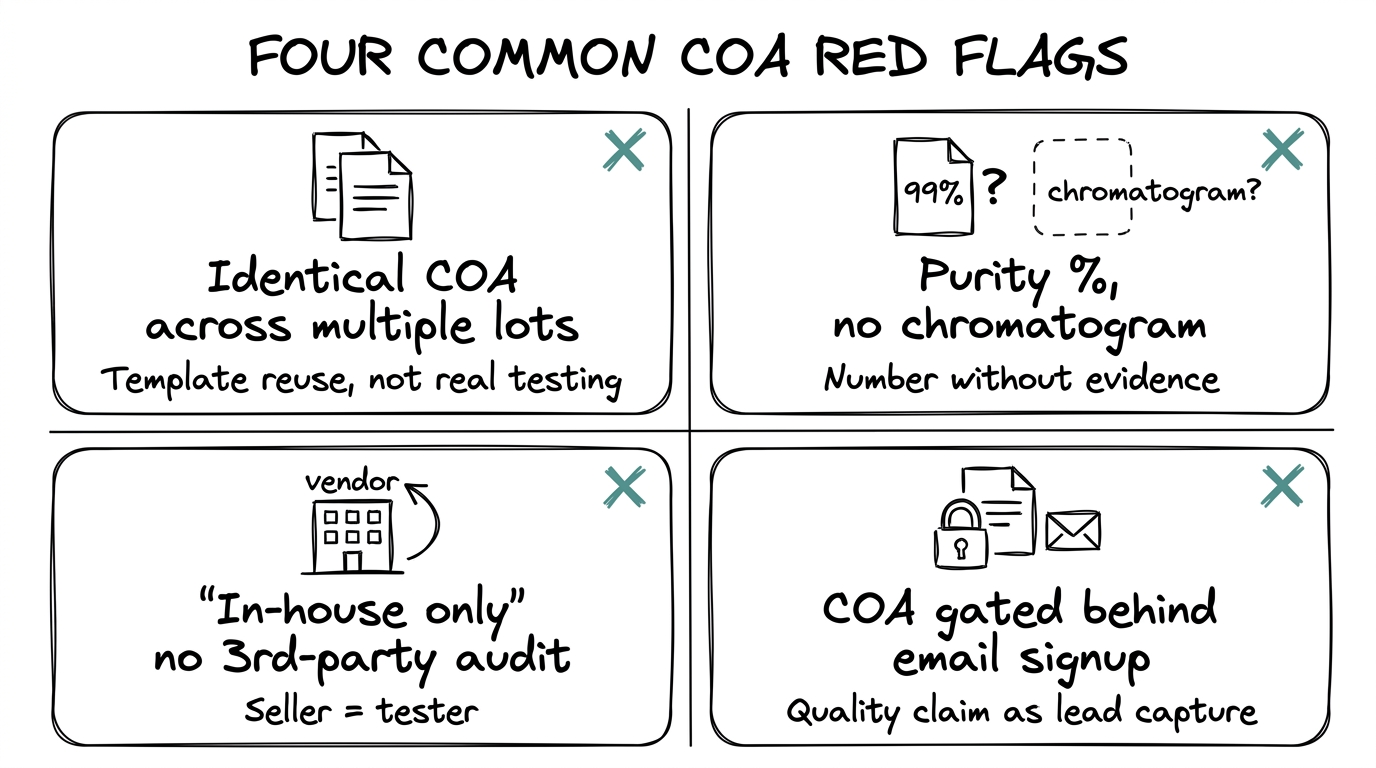
Identical COA across multiple lots
Two batches manufactured weeks apart should not produce byte-for-byte identical test results. When the same document recurs across different lot numbers, the vendor is reusing a template rather than testing each batch.
Purity without a chromatogram
The number alone is unverifiable. The chromatogram is the evidence. A COA that cites purity without the underlying trace is asking to be trusted on a claim the buyer cannot check.
In-house testing with no third-party support
In-house is acceptable when the vendor also commissions periodic third-party audits that the buyer can request. "In-house only" without audit support means the party measuring the product is also the party selling it.
COAs hidden behind signup gates
A real COA should be accessible at the product level, not held back until a buyer hands over contact details. Gating it signals either low confidence in the document or an email-capture play dressed up as quality control.
Example COAs
The fastest way to develop an instinct for COA reading is to look at a few real ones. The BPC-157 and retatrutide product pages both link to current batch-specific COAs that carry the fields described above. Reading the checklist with a real document in front of you is a useful second pass.
For the research-compound context behind those COAs, the BPC-157 research guide covers the mechanism and the tissue types studied, and the retatrutide explainer covers the triple-agonist mechanism and Phase 2 and Phase 3 data.
How this connects to vendor evaluation
Reading a COA well is necessary but not sufficient for evaluating a research-peptide vendor. The full picture is the intersection of documented purity, batch consistency over time, cold-chain integrity in shipping, and honest regulatory framing.
The best peptide vendors guide covers the broader evaluation framework — the COA is one leg of a three-legged stool, not the whole thing.
Quick reference
- ●A COA is batch-specific, not product-specific.
- ●HPLC purity above 95 percent, with a chromatogram attached.
- ●Peptide content is different from purity, and often more useful for dose accuracy.
- ●Mass spec confirms identity; observed mass should match theoretical.
- ●The six-point checklist applies in under a minute.
- ●Identical COAs across lots, missing chromatograms, in-house only without audits, and gated COAs are the four recurring red flags.
Frequently asked questions
What is a peptide Certificate of Analysis?
A COA is a batch-level test report showing the results of analytical testing on a specific manufacturing lot of a peptide. It documents identity, purity, content, water levels, and other quality metrics for that specific batch.
What is the difference between peptide purity and peptide content?
Purity (measured by HPLC) is how clean the peptide fraction is. Content (net peptide) is how much of the total vial mass is actual peptide after accounting for water, counter-ions, and salts. A 10 mg vial with 80% content delivers roughly 8 mg of peptide.
What HPLC purity is considered research grade?
Research-grade peptides should be greater than 95% pure by HPLC. High-grade is 98% or above. The purity percentage should be accompanied by an attached chromatogram showing the peak-area evidence.
How do I verify a peptide COA is legitimate?
Apply the six-point checklist: lot number matches the vial, HPLC purity at or above 95% with chromatogram, mass spec within tolerance, peptide content reported separately, water content reported, and analyst signature with date present.
What are the red flags in a peptide COA?
Four main red flags: identical COA documents across different lot numbers (template reuse), purity reported without a chromatogram, in-house only testing with no third-party audits, and COAs gated behind email signup forms.
What does mass spectrometry confirm on a COA?
Mass spectrometry (ESI-MS) confirms the identity of the compound by measuring its exact molecular mass. The observed mass should match the theoretical mass within a narrow tolerance — a large discrepancy means the vial does not contain the labeled peptide.
What is Karl Fischer titration?
Karl Fischer titration is a chemical test that measures residual moisture in lyophilized peptide powder. Typical range is 1-5%. Excess water suggests incomplete lyophilization and affects both potency and shelf life.
Next step
Verify quality before you order
Every order ships with a batch-specific COA including HPLC chromatogram, mass spec, and peptide content.