Key takeaways
- ●"CJC-1295" usually means the no-DAC version (Mod GRF 1-29) — the with-DAC variant is pharmacologically distinct.
- ●The stack works because two intracellular cascades (cAMP/PKA and calcium/IP3) converge on the same somatotroph for amplified GH release.
- ●Ipamorelin is preferred over GHRP-6 and GHRP-2 because it does not elevate cortisol or prolactin at GH-releasing doses.
- ●The no-DAC version preserves pulsatile GH release that maps onto the natural 4-6 daily pulse pattern.
- ●Standard research protocol is 100 mcg + 100 mcg, 2-3 times daily, subcutaneously.
The short version
CJC-1295 + Ipamorelin is the most-researched growth-hormone peptide stack in the research-peptide space. It shows up in recovery work, body-composition models, age-related GH decline studies, and peptide-receptor pharmacology papers.
The naming is confusing and the mechanism is interesting. This guide covers both.
The stack combines two compounds that hit different receptors on the same pituitary cell. One (Mod GRF 1-29, also called “CJC-1295 no DAC”) activates the GHRH receptor. The other (Ipamorelin) activates the ghrelin receptor. The intracellular cascades converge on the somatotroph and produce amplified, pulsatile growth hormone release that neither compound achieves alone.
The naming clarification first
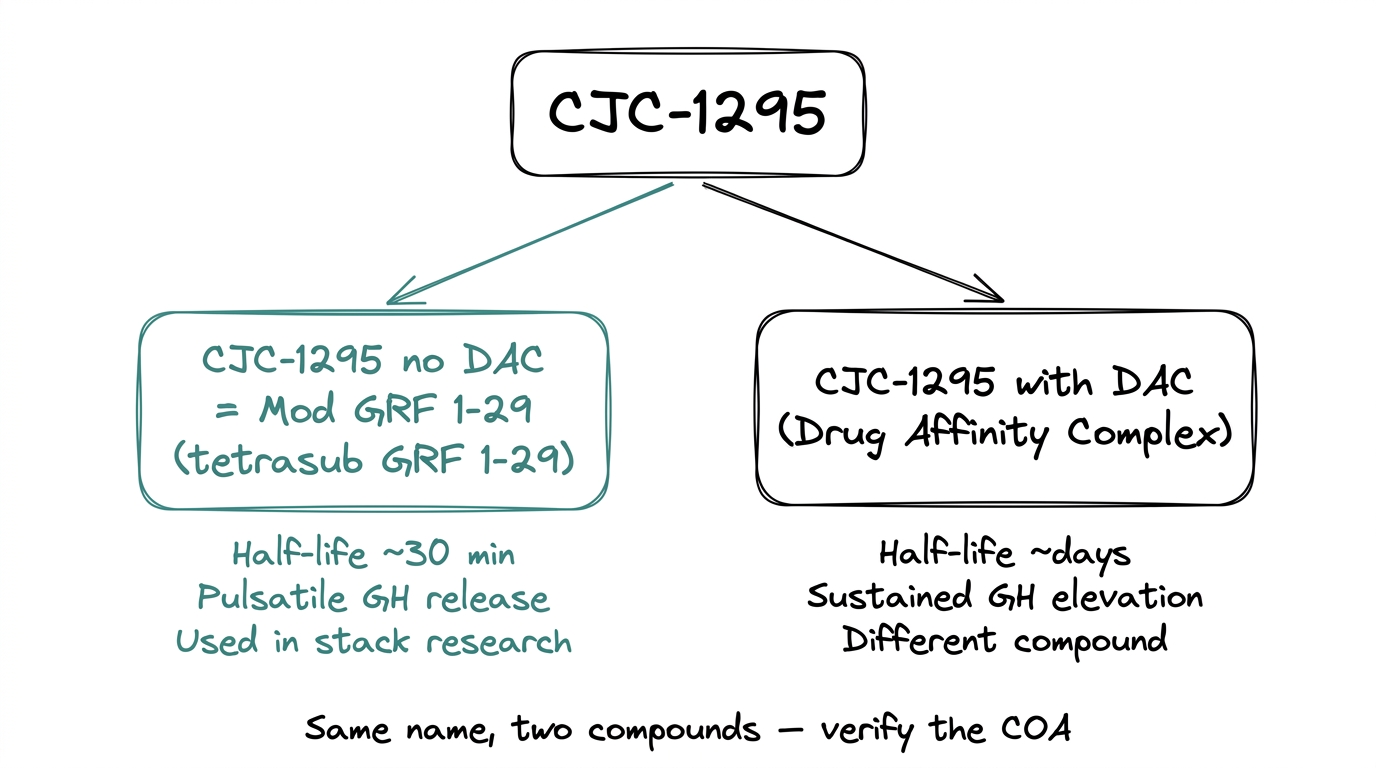
The single most common confusion in this space: “CJC-1295” refers to two related but distinct compounds.
- ●CJC-1295 no DAC (also called Mod GRF 1-29 or tetrasubstituted GRF 1-29). Short-acting. Half-life roughly 30 minutes. Produces pulsatile GH release. This is what people usually mean when they say "CJC-1295 + Ipamorelin."
- ●CJC-1295 with DAC (Drug Affinity Complex). Long-acting. Half-life of days. Produces sustained rather than pulsatile GH elevation.
The two are pharmacologically different. Most of the published stack research uses the no-DAC version. When a product is listed as “CJC-1295” without clarification, check the COA and the product page — the molecular weight and sequence confirm which variant it is.
For this guide, “CJC-1295” means the no-DAC version (Mod GRF 1-29) unless stated otherwise.
Why the two compounds get stacked
The answer is receptor biology. Growth hormone is released from pituitary somatotrophs in response to two different upstream signals, each with its own receptor and intracellular cascade.
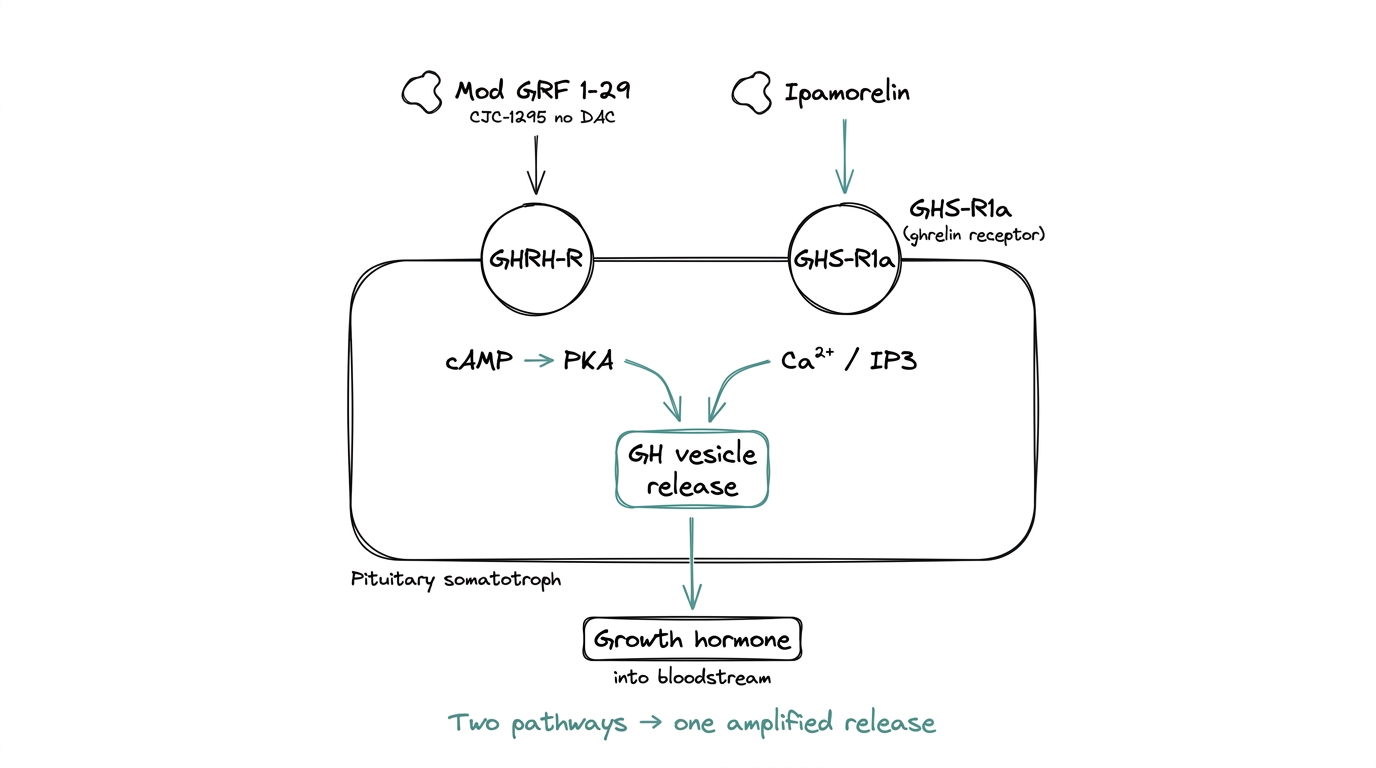
- ●GHRH receptor (GHRH-R). Responds to growth hormone releasing hormone. Signals through the cAMP to PKA pathway. This is the pathway Mod GRF 1-29 targets.
- ●GHS receptor (GHS-R1a, also called the ghrelin receptor). Responds to ghrelin and ghrelin mimetics. Signals through calcium and IP3. This is the pathway Ipamorelin targets.
Activating either receptor alone releases GH. Activating both at once produces a larger release than the sum of the individual activations. The synergy comes from two different intracellular cascades meeting at the same functional endpoint — GH vesicle release — and amplifying each other.
That mechanism is why the stack is more-than-additive, and why it is the default GH-axis research protocol rather than either compound used alone.
Mod GRF 1-29 (the CJC-1295 no-DAC side)
Sequence: GHRH(1-29) with four amino-acid substitutions — D-Ala at position 2, Gln at 8, Ala at 15, Leu at 27. Molecular weight ~3,367 Da. CAS 863288-34-0.
The substitutions serve one purpose: resistance to enzymatic degradation. Native GHRH has a half-life measured in minutes because DPP-IV and other peptidases chew through it quickly. The four substitutions slow that down, extending the half-life to roughly 30 minutes.
That half-life is deliberate. Longer would blur the pulsatile release pattern. Shorter would require impractical dosing frequency. Thirty minutes supports a clean pulsatile profile with two to three injections per research day.
Mechanism: binds GHRH-R on anterior pituitary somatotrophs, activates adenylyl cyclase, raises intracellular cAMP, activates protein kinase A (PKA), and triggers GH release from storage vesicles.
The “with DAC” variant adds a Drug Affinity Complex that binds serum albumin, extending the half-life to multiple days. That produces sustained GH elevation rather than pulsatile — pharmacologically distinct, and typically treated as a separate research variable.
Ipamorelin (the GHS-R side)
Sequence: Aib-His-D-2-Nal-D-Phe-Lys-NH2. A pentapeptide. Molecular weight ~711 Da. CAS 170851-70-4.
Ipamorelin is a ghrelin mimetic. It activates GHS-R1a (the ghrelin receptor), which signals through calcium release and IP3 — a distinct cascade from the GHRH pathway.
The distinguishing feature is selectivity. Earlier GH secretagogues — GHRP-6 and GHRP-2 in particular — activate GHS-R1a but also stimulate ACTH, cortisol, and prolactin release at GH-releasing doses. That creates confounded research data: GH goes up, but so does cortisol, which has its own effects on the downstream variables a researcher usually cares about.
Ipamorelin does not produce significant cortisol or prolactin release at GH-releasing doses. It also does not stimulate appetite the way ghrelin does at typical research doses, despite being a ghrelin mimetic. The receptor interaction is cleaner than the earlier GHRPs.
This selectivity is why Ipamorelin is the GHS-R agonist of choice for stack research, over GHRP-6 or GHRP-2.

| Compound | GH release | Cortisol | Prolactin | Appetite |
|---|---|---|---|---|
| GHRP-6 | Yes | Elevated | Elevated | Elevated |
| GHRP-2 | Strong | Mild increase | Mild increase | Mild increase |
| Ipamorelin | Yes | No change | No change | No change |
How the stack is dosed in research
The standard research protocol is 100 mcg Mod GRF 1-29 + 100 mcg Ipamorelin per administration, typically 2-3 times daily, subcutaneously.
The dosing cadence matters:
- ●Roughly 30-minute half-life on Mod GRF 1-29 drives the 2-3x daily frequency. Longer gaps lose the pulsatility benefit.
- ●Administration is often timed relative to circadian rhythm — research protocols commonly time one injection before sleep, capturing the natural nocturnal GH pulse.
- ●Each injection produces a GH spike within 15-30 minutes that returns to baseline within 2-3 hours.
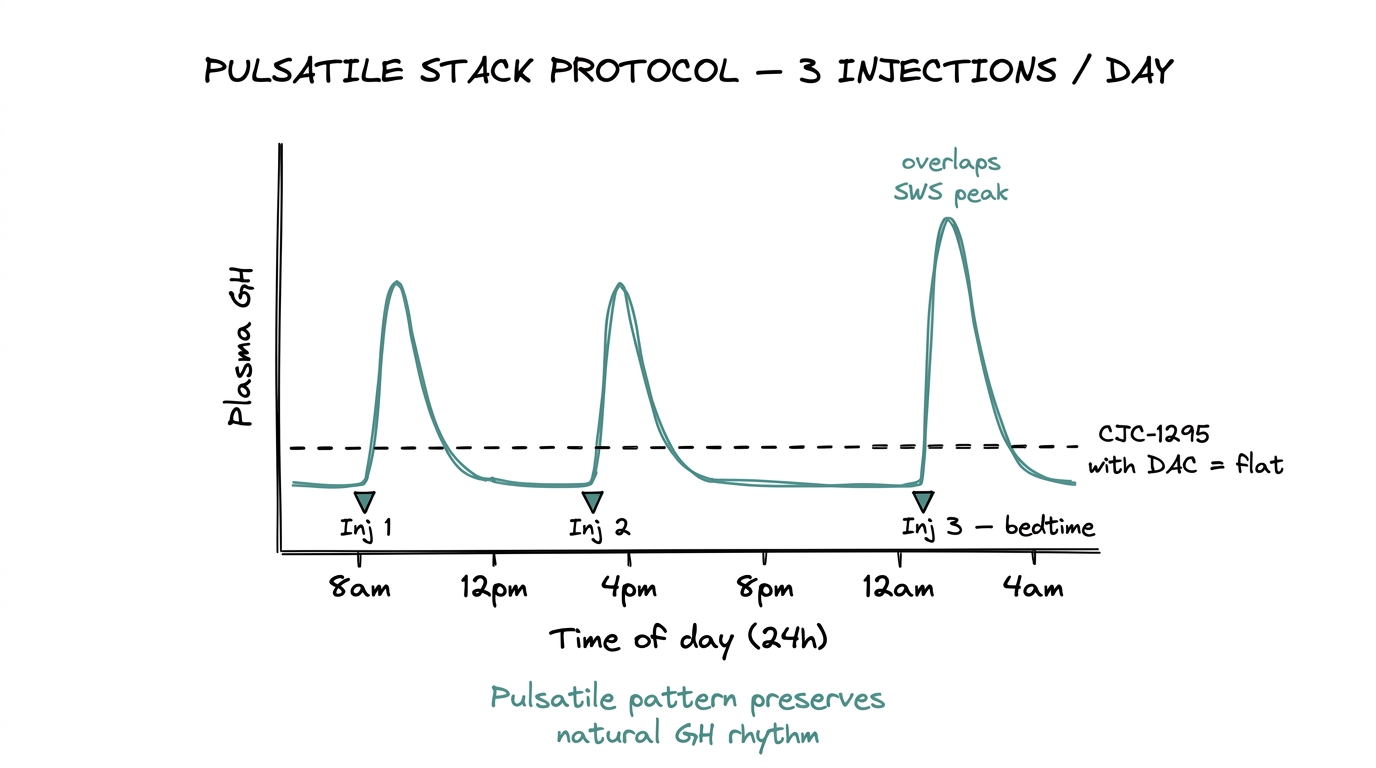
The daily total (3 injections x 100 mcg each compound = 300 mcg each) is modest. The protocol design is about timing and pulsatility, not total dose.
What researchers study with this stack
GH-axis pharmacology
Dual-pathway receptor characterization — comparing GHRH-R alone, GHS-R alone, and the combination, to study the convergence on somatotroph signaling.
Pulsatile vs sustained GH
Comparing the no-DAC stack (pulsatile) to CJC-1295 with DAC (sustained) on downstream variables like lean mass, fat mass, and IGF-1 kinetics.
Age-related GH decline
Older rodents show reduced GH pulse amplitude; the stack is used to study whether external stimulation restores younger pulse patterns.
Body composition
Lean-mass and fat-mass changes in animal models, typically as a secondary outcome after GH and IGF-1 pharmacokinetic characterization.
Sleep architecture
Nocturnal GH pulses map to slow-wave sleep; researchers use the stack to perturb the timing and study sleep-GH coupling.
Receptor selectivity comparisons
Ipamorelin versus GHRP-6 versus GHRP-2 on the same receptor — characterizing the cortisol and prolactin differences.
Most of the literature is preclinical (rodent and some canine models). Human clinical data exists for tesamorelin (a separate GHRH analogue, FDA-approved for HIV-associated lipodystrophy) and for sermorelin (GHRH 1-29), but not for Mod GRF 1-29 or Ipamorelin specifically as approved therapies.
Why the no-DAC version is preferred for stack work
The pulsatility argument is the reason.
Natural GH release is pulsatile. Somatotrophs secrete GH in bursts, typically 4-6 pulses per day in young adults, with the largest pulse occurring during slow-wave sleep. Between pulses, GH drops back to near-baseline. IGF-1 (the downstream output of GH signaling) integrates those pulses into a smoother daily exposure.
CJC-1295 with DAC produces flat, sustained GH elevation — closer to continuous infusion than pulsatile release. That has pharmacologically different consequences, including greater potential for receptor desensitization (somatotrophs downregulate GHRH-R in response to persistent stimulation).
Mod GRF 1-29 (no DAC), stacked with Ipamorelin, produces discrete GH pulses that map onto the natural pulsatile pattern. That is why the no-DAC protocol is the default in GH-axis research, and why the with-DAC version is treated as a separate research variable rather than an equivalent option.
Storage and handling
Both compounds are supplied as lyophilized powder.
| Category | Mod GRF 1-29 | Ipamorelin |
|---|---|---|
| Long-term storage | -20 degrees C | -20 degrees C |
| Short-term storage | 2-8 degrees C | 2-8 degrees C |
| Reconstituted shelf life | 2-3 weeks | 3-4 weeks |
| Stability note | More degradation-sensitive in solution | Relatively stable in solution |
Both should be protected from light and from repeated freeze-thaw cycles. Reconstitution uses bacteriostatic water by default for research work.
Quality signals for stack compounds
For researchers sourcing either compound for stack work, the COA checks apply to each compound independently.
- ●HPLC purity above 98% with a batch-specific Certificate of Analysis for each vial.
- ●Mass spectrometry identity confirmation — ~3,367 Da for Mod GRF 1-29 and ~711 Da for Ipamorelin. The mass spec is how you confirm you have the no-DAC variant specifically, since the with-DAC variant has a different molecular weight.
- ●Sequence verification on the COA. For Mod GRF 1-29, the four substitutions (positions 2, 8, 15, 27) should be explicitly confirmed.
- ●Cold-chain discipline in transit. Both compounds are peptides and both should ship with awareness of thermal exposure.
COA walkthrough
The peptide Certificate of Analysis guide walks through every field on a real COA and the four red flags that signal repackaged or mislabeled material. That walkthrough applies equally to both compounds in the stack.
Limits and tradeoffs
- ●Preclinical dominance. Most of the published stack data is animal work. Human research-grade data on this specific stack is sparse.
- ●Naming confusion in the supply market. "CJC-1295" is used ambiguously across vendors. The no-DAC versus with-DAC distinction is pharmacologically real and worth verifying per batch.
- ●Research-use framing. Both compounds are supplied for research use only. Neither is FDA-approved for human therapeutic use.
- ●Desensitization risk. Sustained activation of either receptor can downregulate receptor expression. The pulsatile protocol is designed around that, but longer-duration studies should account for it.
Where to go from here
For the broader question of how to verify either compound's quality before use, the peptide Certificate of Analysis guide covers the six-point checklist and the four red flags that signal a weak supply. For the framework for evaluating any vendor carrying these compounds, the best peptide vendors 2026 framework covers the five-criteria evaluation.
Quick reference
- ●"CJC-1295" usually means the no-DAC version = Mod GRF 1-29. The with-DAC version is a different compound with a different half-life.
- ●The stack hits two receptors on the same cell: GHRH-R (cAMP/PKA) and GHS-R1a (calcium/IP3). Synergy comes from cascade convergence.
- ●Ipamorelin's advantage over GHRP-6/GHRP-2 is selectivity — no meaningful cortisol or prolactin bump at GH-releasing doses.
- ●Typical research protocol: 100 mcg + 100 mcg, 2-3x daily, subcutaneous.
- ●No-DAC is preferred in stack research because it preserves pulsatile GH release; with-DAC produces flat sustained exposure.
- ●COA checks: HPLC above 98%, mass spec identity (~3,367 Da and ~711 Da), sequence verification, cold-chain discipline.
Frequently asked questions
What is CJC-1295 no DAC?
CJC-1295 no DAC is also called Mod GRF 1-29. It is a modified GHRH fragment with a ~30-minute half-life that produces pulsatile growth hormone release. It is pharmacologically distinct from CJC-1295 with DAC, which has a half-life of days.
Why are CJC-1295 and Ipamorelin used together?
They activate two different receptors (GHRH-R and GHS-R1a) on the same pituitary somatotroph cell. The two intracellular cascades converge and produce a GH release that is greater than the sum of either compound alone.
What is the standard research dose for the CJC-1295 + Ipamorelin stack?
The standard research protocol is 100 mcg of each compound per administration, typically 2-3 times daily via subcutaneous injection.
Why is Ipamorelin preferred over GHRP-6 or GHRP-2?
Ipamorelin is selective — it releases GH without significantly elevating cortisol, prolactin, or appetite at GH-releasing doses. GHRP-6 and GHRP-2 elevate all three, which confounds research data.
What is the difference between CJC-1295 with DAC and without DAC?
The no-DAC version (Mod GRF 1-29) has a ~30-minute half-life and produces pulsatile GH release. The with-DAC version binds serum albumin, extending the half-life to days and producing sustained rather than pulsatile GH elevation.
How should CJC-1295 and Ipamorelin be stored?
Both should be stored lyophilized at -20 degrees C long-term or 2-8 degrees C short-term. Once reconstituted with bacteriostatic water, Mod GRF 1-29 should be used within 2-3 weeks and Ipamorelin within 3-4 weeks.
Is the CJC-1295 + Ipamorelin stack FDA-approved?
No. Neither Mod GRF 1-29 nor Ipamorelin is FDA-approved for human therapeutic use. Both are supplied for research use only.
Next step
Source research-grade GH-axis compounds
Each vial ships with a batch-specific COA including HPLC purity and mass spectrometry identity confirmation.