Key takeaways
- ●Retatrutide is a triple agonist activating GLP-1, GIP, and glucagon receptors with a single molecule.
- ●Phase 2 showed 24.2% body weight reduction at 48 weeks; TRIUMPH-4 Phase 3 showed 28.7% at 68 weeks.
- ●The glucagon receptor component drives hepatic fat oxidation, supported by Phase 2a MASH trial data.
- ●Dysesthesia is a new dose-dependent safety signal from TRIUMPH-4 that was absent in Phase 2.
- ●FDA approval estimated for 2027-2028 if the seven-trial TRIUMPH program succeeds.
Retatrutide is a single synthetic peptide that activates three metabolic receptors at once — GLP-1, GIP, and glucagon — and it is currently the highest-profile compound in the post-semaglutide weight-research wave.
One molecule, three receptor systems, which is why the trial data has drawn so much attention.
Origin and development
Eli Lilly developed retatrutide under the internal code LY3437943, starting from the same GLP-1 scaffold that made semaglutide and tirzepatide successful. Phase 2 obesity data published in the New England Journal of Medicine in 2023 showed dose-dependent body weight reduction up to 24.2% at 48 weeks. That number exceeded semaglutide (~15%) and tirzepatide (~20%) at comparable timepoints and made retatrutide the most-watched compound in the category.
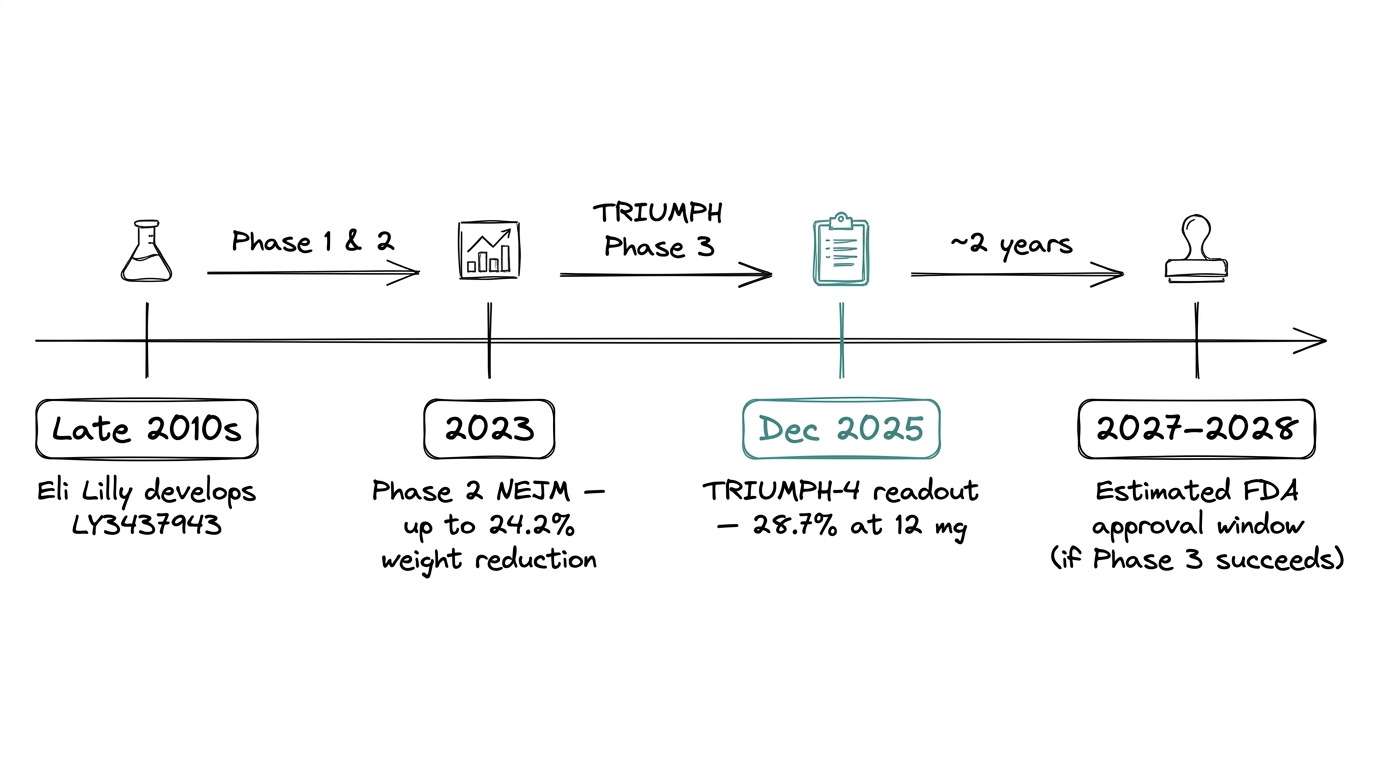
The Phase 3 TRIUMPH program is currently running across seven trials. The first readout, TRIUMPH-4 in December 2025, reported 28.7% mean body weight reduction at 68 weeks in the 12 mg cohort. Approval is estimated for 2027 to 2028 if the full program succeeds.
How it works
Retatrutide is a triple agonist. Two concepts to unpack.
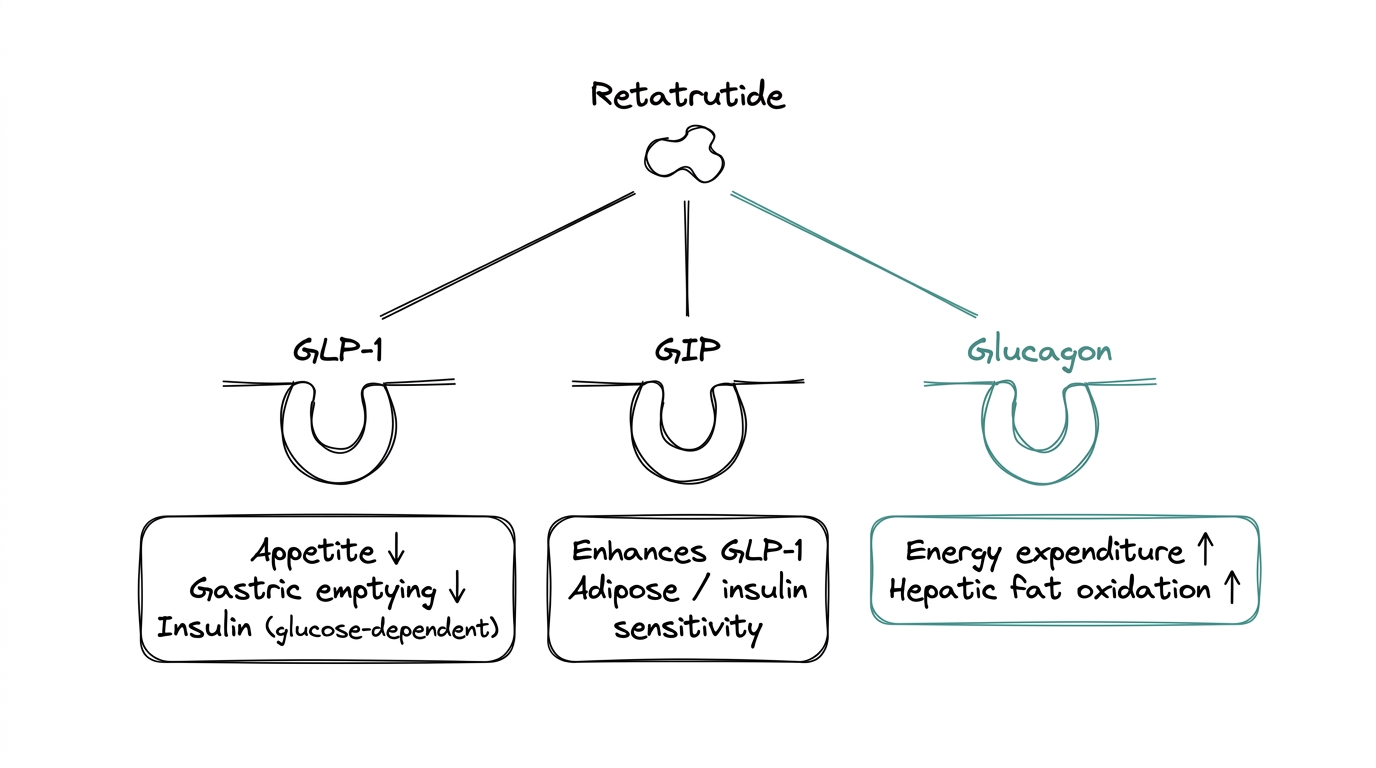
A receptor is a docking site on a cell that responds to a specific signal. An agonist is a molecule that activates a receptor the same way the body's natural signal would. Retatrutide activates three different receptors with one molecule, each one driving a different metabolic lever.
GLP-1 receptor agonism
GLP-1 stands for glucagon-like peptide-1, an incretin hormone the gut releases after eating. Activating its receptor reduces appetite, slows gastric emptying, and triggers glucose-dependent insulin release. This is the same pathway semaglutide uses. Well characterized, well understood, mechanism established for more than a decade.
GIP receptor agonism
GIP stands for glucose-dependent insulinotropic polypeptide, a second incretin hormone released alongside GLP-1. Its independent role is still debated, but tirzepatide — which activates GLP-1 and GIP — outperformed semaglutide (GLP-1 alone) in head-to-head comparisons, which validated GIP as a meaningful additive lever.
Glucagon receptor agonism
This is the distinguishing feature of retatrutide and the counterintuitive one. Glucagon normally raises blood glucose, which is the opposite of what a metabolic research compound usually aims for. Activating the glucagon receptor increases energy expenditure and drives hepatic fat oxidation — the liver burning its own fat stores. The working hypothesis is that the GLP-1 and GIP components offset the glucose-raising effect while letting the energy-expenditure benefit come through.
The result is one peptide covering three metabolic levers: less food intake (GLP-1), enhanced insulin response (GLP-1 + GIP), and increased energy burn (glucagon).
What the trial data shows
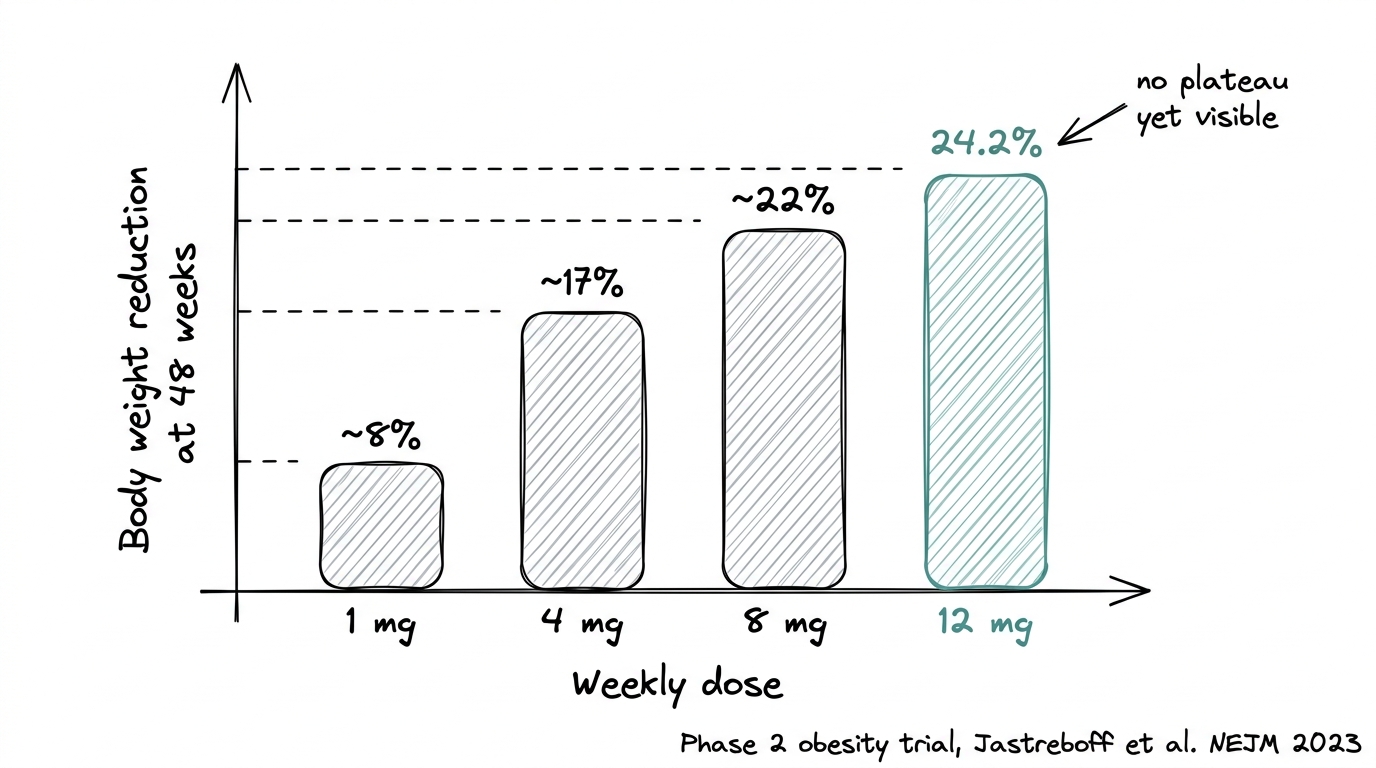
The Phase 2 dose-response was unusually clean. Each higher dose produced measurably more weight reduction, with the 12 mg cohort reaching 24.2% at 48 weeks and no plateau yet visible. That dose-response signal is part of what pushed Lilly into a seven-trial Phase 3 program.
The Phase 2a MASH trial, published in Nature Medicine in 2024, showed significant reductions in liver fat content, supporting the glucagon-component hypothesis — that the unusual receptor choice is what drives hepatic fat oxidation beyond what GLP-1 can do alone.
TRIUMPH-4 in December 2025 reported the first Phase 3 numbers: 28.7% mean body weight reduction at 68 weeks at 12 mg, with 58.6% of participants achieving at least 25% total body weight loss. WOMAC pain scores in the knee-osteoarthritis cohort decreased by up to 75.8%. Non-HDL cholesterol, triglycerides, hsCRP, and systolic blood pressure all dropped at the highest dose.
New safety signal
A new safety signal appeared in TRIUMPH-4. Dysesthesia, a skin-sensation side effect, was reported in 8.8% of the 9 mg group and 20.9% of the 12 mg group. It was absent from the Phase 2 data. That signal will shape dose conversations in the rest of the TRIUMPH program.
How it compares
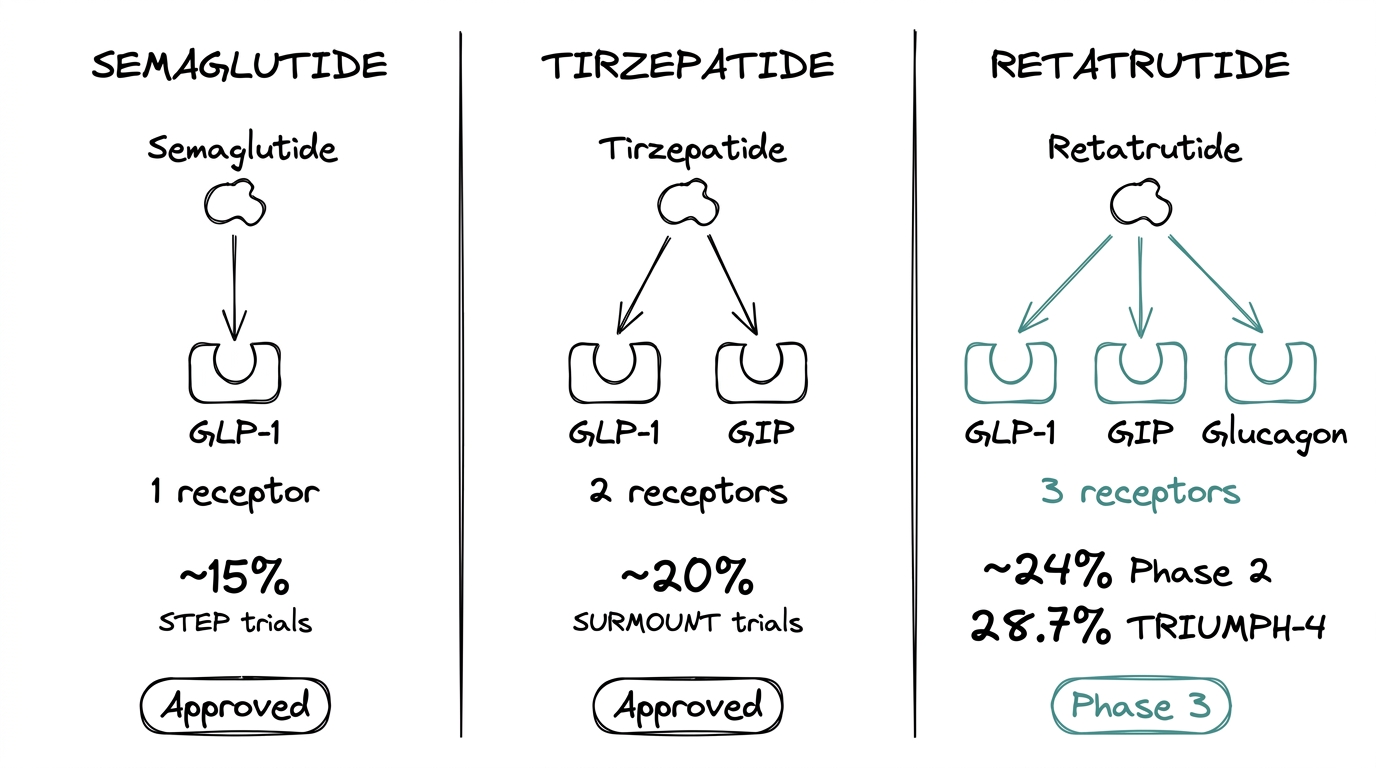
| Semaglutide | Tirzepatide | Retatrutide | |
|---|---|---|---|
| Receptors | GLP-1 | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Weight reduction | ~15% | ~20% | ~24% (Ph2), 28.7% (Ph3) |
| Status | Approved | Approved | Phase 3 |
The side-by-side is the clearest way to see what the third receptor buys. Semaglutide covers one receptor. Tirzepatide covers two. Retatrutide covers three. The published weight-reduction numbers follow that progression.
The right compound depends on the question being asked. More receptors means more downstream effects to track and more interactions to characterize. For a narrow GLP-1 question, semaglutide is cleaner. For a question about combined incretin and glucagon signaling, retatrutide is the only compound that covers the full range.
What researchers are exploring
Research interest in retatrutide clusters around three questions.
Can the glucagon receptor be useful without raising blood glucose?
The glucagon arm is the novel bet. If the GLP-1 plus GIP components fully offset glucagon's glycaemic effect, the triple-agonist approach opens new ground in metabolic research. The Phase 2a MASH data supports the hypothesis, but the mechanism is still being mapped.
How does triple agonism behave over longer timeframes?
Phase 2 data runs to 48 weeks. TRIUMPH-4 runs to 68 weeks. The longer-duration readouts in the rest of the TRIUMPH program will show whether the weight curve plateaus, holds, or continues.
Where does it sit relative to tirzepatide head-to-head?
Tirzepatide is the current commercial benchmark. Research protocols comparing retatrutide directly against tirzepatide at matched doses and timepoints will clarify how much of the advantage comes from the glucagon component versus the higher dose ceiling.
Limits and tradeoffs
Retatrutide is Phase 3 investigational. Approval for human therapeutic use is not yet granted, and research-use sourcing is the only legitimate channel in most jurisdictions.
The dysesthesia signal from TRIUMPH-4 is worth watching. It is dose-dependent and was invisible in the smaller Phase 2 dataset — a reminder that Phase 2 safety data is preliminary.
The compound is larger than semaglutide at roughly 4700 Da and 39 amino acids. Handling during reconstitution takes more care, and cold-chain discipline matters more than for smaller peptides.
Retatrutide is also expensive at current supply, partly because synthesis is more complex and partly because demand is outrunning capacity while Phase 3 runs.
Where to go from here
The leading triple-agonist compound
Retatrutide is the leading compound for research questions that need more than GLP-1. For GLP-1-only questions, semaglutide is the reference point. For GLP-1 plus GIP, tirzepatide is the approved option. For the full triple agonism, retatrutide is the only compound covering all three.
For a direct comparison against the most familiar reference, see the retatrutide vs ozempic guide.
Frequently asked questions
What is retatrutide?
Retatrutide (LY3437943) is a synthetic peptide developed by Eli Lilly that activates three metabolic receptors at once: GLP-1, GIP, and glucagon. It is currently in Phase 3 clinical trials.
What makes retatrutide different from semaglutide?
Semaglutide activates only the GLP-1 receptor. Retatrutide activates GLP-1, GIP, and glucagon receptors — three metabolic levers versus one. The Phase 2 weight-reduction numbers reflect that difference: ~24% for retatrutide versus ~15% for semaglutide.
What is a triple agonist?
A triple agonist is a single molecule that activates three different receptors. Retatrutide activates GLP-1 (appetite and insulin), GIP (incretin enhancement), and glucagon (energy expenditure and hepatic fat oxidation).
What did the TRIUMPH-4 trial show?
TRIUMPH-4 reported 28.7% mean body weight reduction at 68 weeks at 12 mg, with 58.6% of participants achieving at least 25% total body weight loss. It also revealed a new dysesthesia safety signal at higher doses.
Is retatrutide FDA approved?
No. Retatrutide is in Phase 3 clinical trials. FDA approval is estimated for 2027 to 2028 if the TRIUMPH program succeeds. It is currently available for research use only.
Why does retatrutide include glucagon receptor activation?
The glucagon receptor increases energy expenditure and drives hepatic fat oxidation. The GLP-1 and GIP components are hypothesized to offset glucagon's glucose-raising effect while preserving the energy-burn benefit.
What is the dysesthesia signal in TRIUMPH-4?
Dysesthesia is a skin-sensation side effect reported in 8.8% of the 9 mg group and 20.9% of the 12 mg group. It was absent from the smaller Phase 2 dataset and is dose-dependent.
Next step
Explore metabolic research peptides
Research-grade compounds with batch-specific COAs and cold-chain shipping.