Key takeaways
- ●Semaglutide (Ozempic) activates one receptor (GLP-1); retatrutide activates three (GLP-1, GIP, glucagon).
- ●The Phase 3 weight-reduction delta is ~14 percentage points: 14.9% for semaglutide vs 28.7% for retatrutide at matched 68-week timepoints.
- ●Ozempic is FDA-approved; retatrutide is Phase 3 investigational with estimated approval in 2027-2028.
- ●The glucagon receptor component drives hepatic fat oxidation and energy expenditure — the mechanism that separates retatrutide from dual agonists.
- ●Quality verification matters more for newer, less-commoditized compounds like retatrutide.
The short version
Ozempic is the brand name for semaglutide — a single-receptor GLP-1 agonist approved for type 2 diabetes and weight management. Retatrutide is an investigational triple agonist (GLP-1, GIP, and glucagon) currently in Phase 3. The comparison keeps coming up because retatrutide is the compound most often framed as “the next one after Ozempic.”
This guide covers the mechanism difference, the trial data that separates them, and how to think about each one for research purposes.
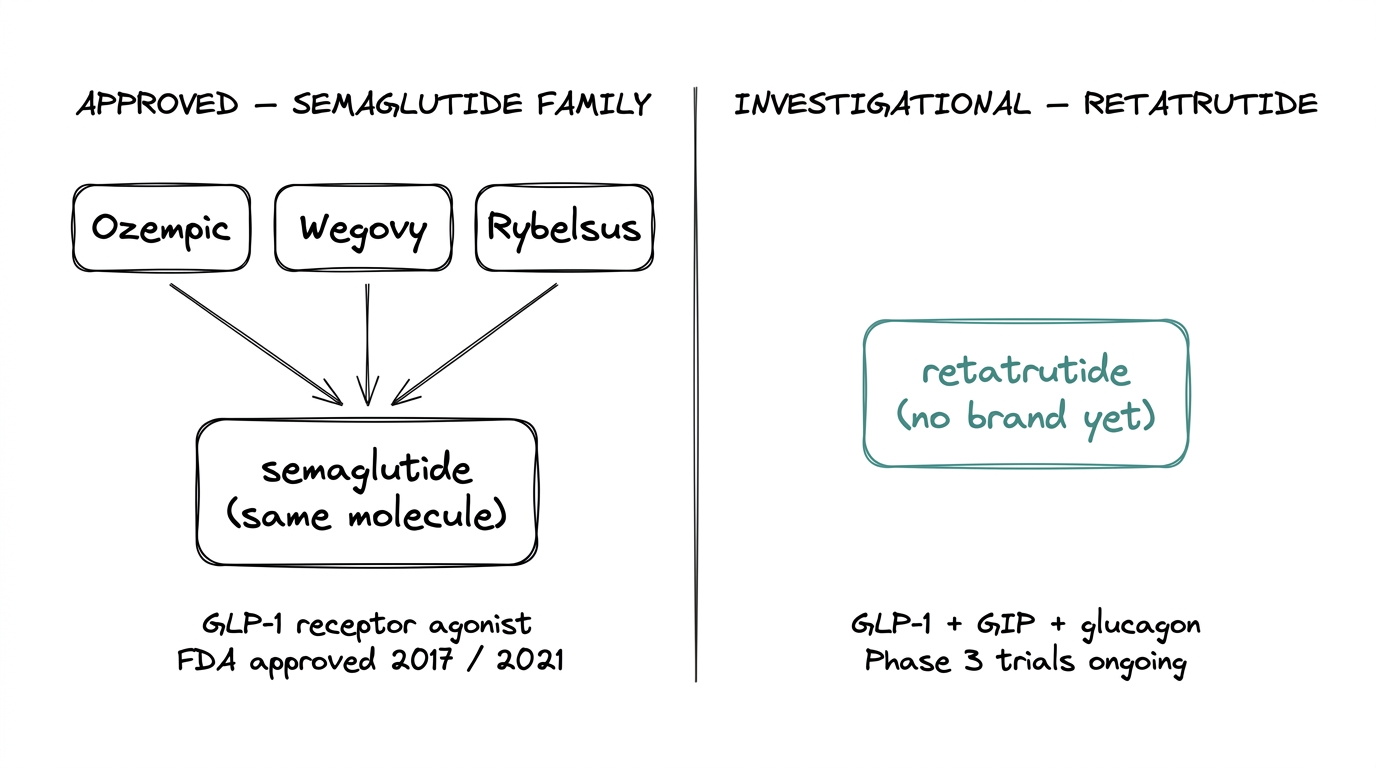
- ●Ozempic = semaglutide. Same molecule, different brand packaging. Wegovy is semaglutide at a higher dose for weight management. Rybelsus is oral semaglutide. All three are the same compound.
- ●Semaglutide activates one receptor (GLP-1). Phase 3 trials showed roughly 15% body weight reduction in obesity indications.
- ●Retatrutide activates three receptors (GLP-1, GIP, glucagon). Phase 2 showed up to 24.2% body weight reduction. Phase 3 TRIUMPH-4 showed 28.7% at 68 weeks at the 12 mg dose.
- ●Ozempic is approved. Retatrutide is not. Approval for retatrutide is estimated for 2027-2028 pending full TRIUMPH program readout.
The retatrutide explainer covers the triple-agonist mechanism in full detail, and the semaglutide explainer covers the GLP-1 reference profile.
Why the comparison exists
Semaglutide defined the current metabolic-peptide wave. When people started asking “what is the upgrade?”, retatrutide was the answer that generated the most attention — because it is the first compound to activate three metabolic receptors at once, not just one or two.
The question behind the comparison is usually: does the third receptor buy anything real, or is it just higher numbers at higher doses? The data so far says the third receptor buys something. How much depends on how you count it.
Mechanism side-by-side
Ozempic (semaglutide) mechanism
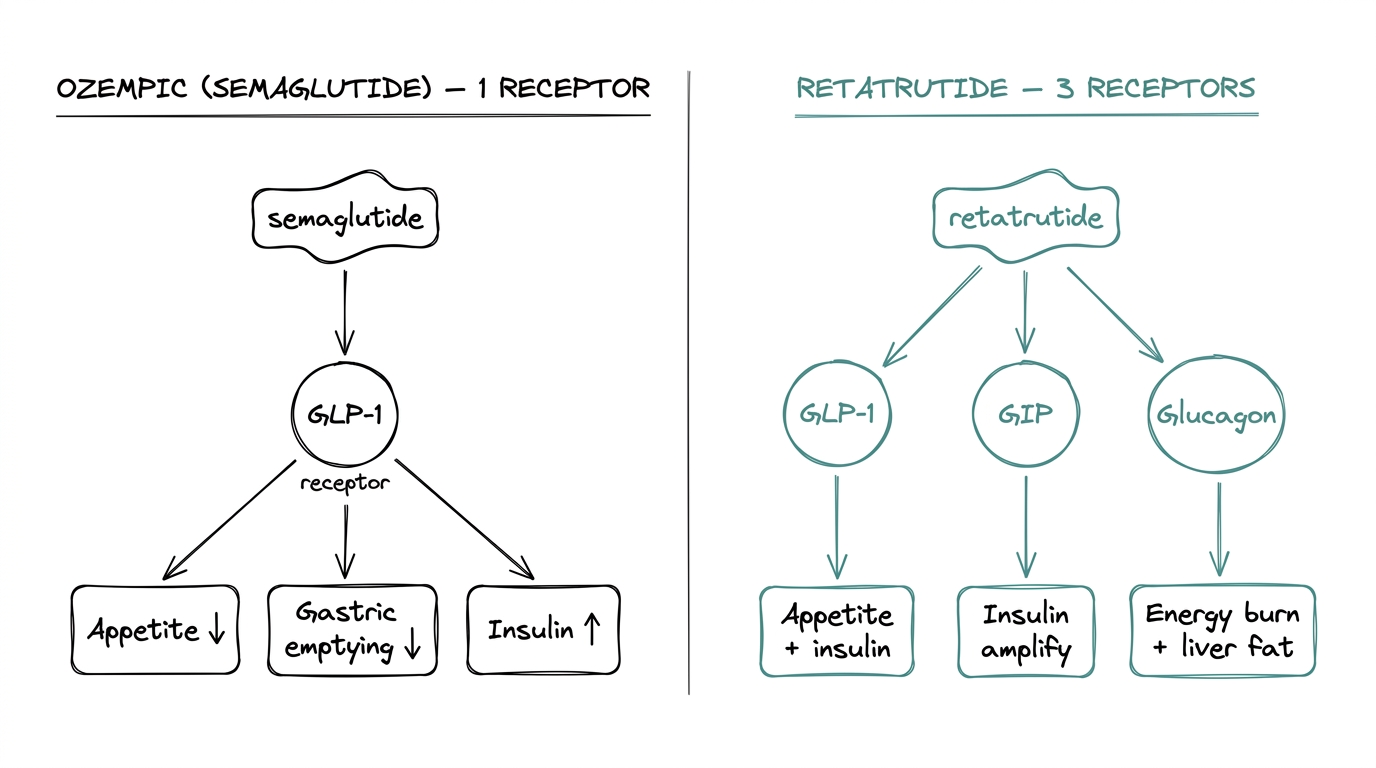
Semaglutide is a single-receptor agonist. It activates the GLP-1 receptor, which is the docking site for glucagon-like peptide-1 — an incretin hormone released after eating. Activating this one receptor drives three downstream effects: reduced appetite, slower gastric emptying, and glucose-dependent insulin release.
One receptor, three downstream outcomes. Mechanism well characterized, clinical data deep.
Retatrutide mechanism
Retatrutide activates three receptors from a single molecule:
- ●GLP-1 receptor. Same pathway as semaglutide. Appetite, gastric emptying, insulin.
- ●GIP receptor. Glucose-dependent insulinotropic polypeptide. A second incretin hormone released alongside GLP-1. Tirzepatide validated GIP as a meaningful additive lever when it outperformed semaglutide in head-to-head comparisons.
- ●Glucagon receptor. The counterintuitive one. Glucagon normally raises blood glucose, which is the opposite of what a metabolic research compound usually aims for. Activating this receptor increases energy expenditure and drives hepatic fat oxidation — the liver burning its own fat stores. The working hypothesis is that GLP-1 and GIP components offset the glucose-raising effect while the energy-burn benefit comes through.
Three receptors, covering three metabolic levers at once: less food intake (GLP-1), enhanced insulin response (GLP-1 + GIP), and increased energy burn (glucagon).
What the trial data shows
Numbers from the published trials put the compounds in direct relationship.
Semaglutide — STEP program (Phase 3)
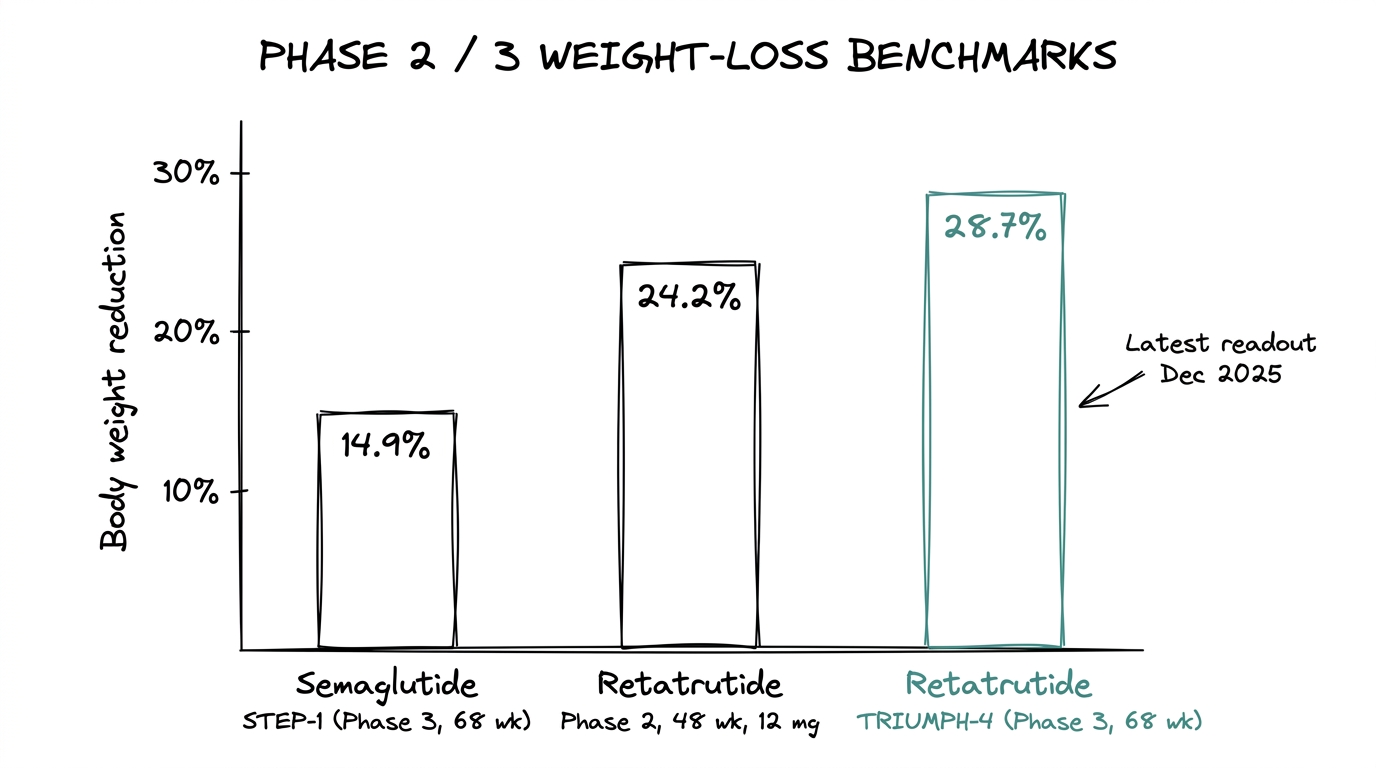
14.9% mean body weight reduction at 68 weeks on 2.4 mg weekly (STEP-1, 2021). Extensively replicated across follow-up trials. This is the benchmark number that every new metabolic compound gets compared against.
Retatrutide — Phase 2 obesity
Dose-dependent reduction: ~8% at 1 mg, ~17% at 4 mg, ~22% at 8 mg, 24.2% at 12 mg, all at 48 weeks. The curve keeps rising without a visible plateau. (Jastreboff et al., NEJM 2023.)
Retatrutide — TRIUMPH-4 Phase 3
28.7% mean body weight reduction at 68 weeks in the 12 mg cohort. 58.6% of participants achieved at least 25% total body weight loss. WOMAC knee-pain scores decreased by up to 75.8% in the osteoarthritis cohort. A dysesthesia signal appeared — 8.8% at 9 mg, 20.9% at 12 mg — that was not visible in the smaller Phase 2 dataset. (December 2025.)
The raw delta is roughly 14 percentage points between the established Ozempic Phase 3 number and the TRIUMPH-4 retatrutide Phase 3 number at matched 68-week timepoints. That is the single cleanest data point separating them.
Regulatory status
- ●Ozempic / Wegovy: FDA-approved. Ozempic for type 2 diabetes (2017) and Wegovy for weight management (2021). Both are semaglutide at different doses.
- ●Retatrutide: Phase 3 investigational. No approval yet. Estimated 2027-2028 if the full TRIUMPH program succeeds.
This is the hardest practical difference. Ozempic is a prescribed therapy accessible through standard pharmacy channels. Retatrutide is only available through research-use-only supply until the approval window opens.
Pharmacokinetics and dosing
Both compounds are weekly injections. Both were designed for that cadence intentionally — a weekly injectable is the clinical sweet spot for adherence in metabolic indications.
Semaglutide half-life is roughly 7 days. Retatrutide half-life is similar, also supporting once-weekly dosing.
The molecular weights differ. Semaglutide is ~4,100 Da. Retatrutide is ~4,700 Da. The larger peptide requires slightly more care in reconstitution and cold-chain handling.
Safety comparison
| Category | Ozempic (semaglutide) | Retatrutide |
|---|---|---|
| Safety data depth | Years of post-approval surveillance | Phase 3 — still being characterized |
| GI side effects | Nausea, diarrhea, constipation — well documented | Similar GI profile to other incretin mimetics |
| Thyroid signal | Black-box warning from rodent carcinogenicity | Not yet characterized at scale |
| Dysesthesia | Not a significant signal | 8.8% at 9 mg, 20.9% at 12 mg (TRIUMPH-4) |
| Pancreatitis | Rare signal | Not yet fully characterized |
The short version: Ozempic's safety profile is broad and well-mapped. Retatrutide's safety profile is narrower and still being filled in — which is normal for a Phase 3 compound.
Cost and access
Ozempic runs $900-1,200 per month in the US at list price. Wegovy runs higher. Insurance coverage is uneven for weight-management indications.
Retatrutide, because it is pre-approval, is only available as research-grade supply for research-use-only work. Pricing varies with supply, and the compound is more expensive than semaglutide partly because synthesis is more complex (larger molecule, triple-agonist design) and partly because Phase 3 demand is running ahead of supply capacity.
When each one fits
The question-matching approach is cleaner than “which is better.”
Reach for Ozempic (semaglutide) when
The research question is about GLP-1 receptor biology specifically, the work benefits from extensive published clinical data, or the comparison baseline is an approved therapy.
Reach for retatrutide when
The research question touches GIP or glucagon receptor biology, the hypothesis involves hepatic fat oxidation or energy expenditure beyond what GLP-1 alone explains, or the goal is to characterize the frontier of combined incretin and glucagon agonism.
Stay with the approved compound when
The study design needs a well-characterized reference. Semaglutide is that reference. Retatrutide is the new variable.
Limits and tradeoffs
Both compounds share the same practical limits. Injection site reactions, GI side effects, and the standard caveats of research-use-only supply for the retatrutide case. Neither is an anti-aging compound or a performance-enhancing compound in any serious sense — both are metabolic tools with narrow, well-defined receptor targets.
Research-use-only framing matters more for retatrutide than for semaglutide. The approved compound has a human therapeutic use case and a clear regulatory home. The investigational compound does not yet, and research protocols need to treat it accordingly.
Quality signals for retatrutide supply
For researchers sourcing retatrutide for research work, three things determine whether the material is fit for purpose:
- ●HPLC purity above 98% with a batch-specific Certificate of Analysis.
- ●Mass spectrometry identity confirmation showing the ~4,700 Da molecular weight.
- ●Cold-chain discipline in transit. Lyophilized retatrutide is stable short-term at ambient but should be stored at -20 degrees C long-term.
COA verification matters more for newer compounds
The peptide Certificate of Analysis guide walks through every field on a real COA and the red flags that signal repackaged or mislabeled material — which matters more for the newer and less-commoditized compound.
Where to go from here
For the full mechanism and trial data on retatrutide alone, the retatrutide explainer covers the compound end-to-end. For the broader question of how to evaluate any research-peptide vendor, the best peptide vendors framework covers the five-criteria evaluation for sourcing either compound.
Quick reference
- ●Ozempic = semaglutide (brand name). Wegovy and Rybelsus are also semaglutide at different doses/routes.
- ●Semaglutide: single GLP-1 receptor. ~15% body weight reduction at Phase 3.
- ●Retatrutide: triple receptor (GLP-1, GIP, glucagon). 24.2% Phase 2, 28.7% TRIUMPH-4.
- ●Ozempic is approved. Retatrutide is Phase 3 investigational.
- ●Both are weekly injections. Retatrutide is the larger molecule and is more expensive at current supply.
- ●The delta between the two Phase 3 numbers is the cleanest single data point: ~14 percentage points.
Frequently asked questions
Is retatrutide the same as Ozempic?
No. Ozempic is the brand name for semaglutide, a single GLP-1 receptor agonist. Retatrutide is a separate investigational compound that activates three receptors (GLP-1, GIP, and glucagon).
How much more weight loss does retatrutide produce compared to Ozempic?
In Phase 3 trials at matched 68-week timepoints, semaglutide showed ~14.9% body weight reduction (STEP-1) while retatrutide showed 28.7% (TRIUMPH-4 at 12 mg) — a delta of roughly 14 percentage points.
Is retatrutide FDA-approved?
No. Retatrutide is currently in Phase 3 clinical trials. Approval is estimated for 2027-2028 pending the full TRIUMPH program readout.
What does the glucagon receptor add in retatrutide?
The glucagon receptor component increases energy expenditure and drives hepatic fat oxidation — the liver burning its own fat stores — while the GLP-1 and GIP components offset the glucose-raising effect of glucagon activation.
Are Ozempic, Wegovy, and Rybelsus the same drug?
Yes. All three are semaglutide at different doses and routes of administration. Ozempic is for type 2 diabetes, Wegovy is for weight management at a higher dose, and Rybelsus is the oral formulation.
What are the side effects of retatrutide vs Ozempic?
Both share GI side effects (nausea, diarrhea, constipation). Retatrutide showed a dose-dependent dysesthesia signal in TRIUMPH-4 (8.8% at 9 mg, 20.9% at 12 mg) that was not seen with semaglutide. Semaglutide has a thyroid-cancer black-box warning from rodent studies.
How often are retatrutide and Ozempic injected?
Both are weekly injections. Both have half-lives of approximately 7 days, supporting once-weekly dosing.
Next step
Explore research-grade metabolic compounds
Compare the metabolic peptide line with batch-specific COAs and transparent quality documentation.