Key takeaways
- ●Most peptide research problems are handling problems, not sourcing problems — correct technique matters as much as correct sourcing.
- ●Reconstitute with bacteriostatic water down the vial wall (never directly onto powder) and never shake.
- ●Lyophilized peptides store for years; reconstituted solutions last 2-4 weeks with bacteriostatic water or 24-48 hours with sterile water.
- ●Run the two-step dosing math (concentration, then dose volume) once per vial and write it down.
- ●Track three dates per vial: manufacture, reconstitution, and use-by.
Before the vial arrives
Most problems with research-grade peptides are handling problems.
A correctly sourced peptide handled badly looks identical to a badly sourced peptide handled correctly. The research data suffers either way.
This guide covers the basics: reconstitution, storage, dosing math, and post-reconstitution shelf life.
Three things should be ready before the package opens:
- ●Bacteriostatic water (0.9% benzyl alcohol sterile water). Regular sterile water works for same-day use. Bacteriostatic water is what extends post-reconstitution shelf life.
- ●Sterile syringes appropriate to the dose volume being drawn. Insulin syringes for small volumes, larger syringes for larger volumes.
- ●Alcohol swabs and a clean work surface — any clean, dry, uncluttered space works.
Having this ready before the vial arrives means the vial spends less time at unstable temperatures between delivery and storage.
Reconstitution
Reconstitution is the process of turning a lyophilized (freeze-dried) peptide powder into a liquid that can be measured and used.
Six steps, in order:
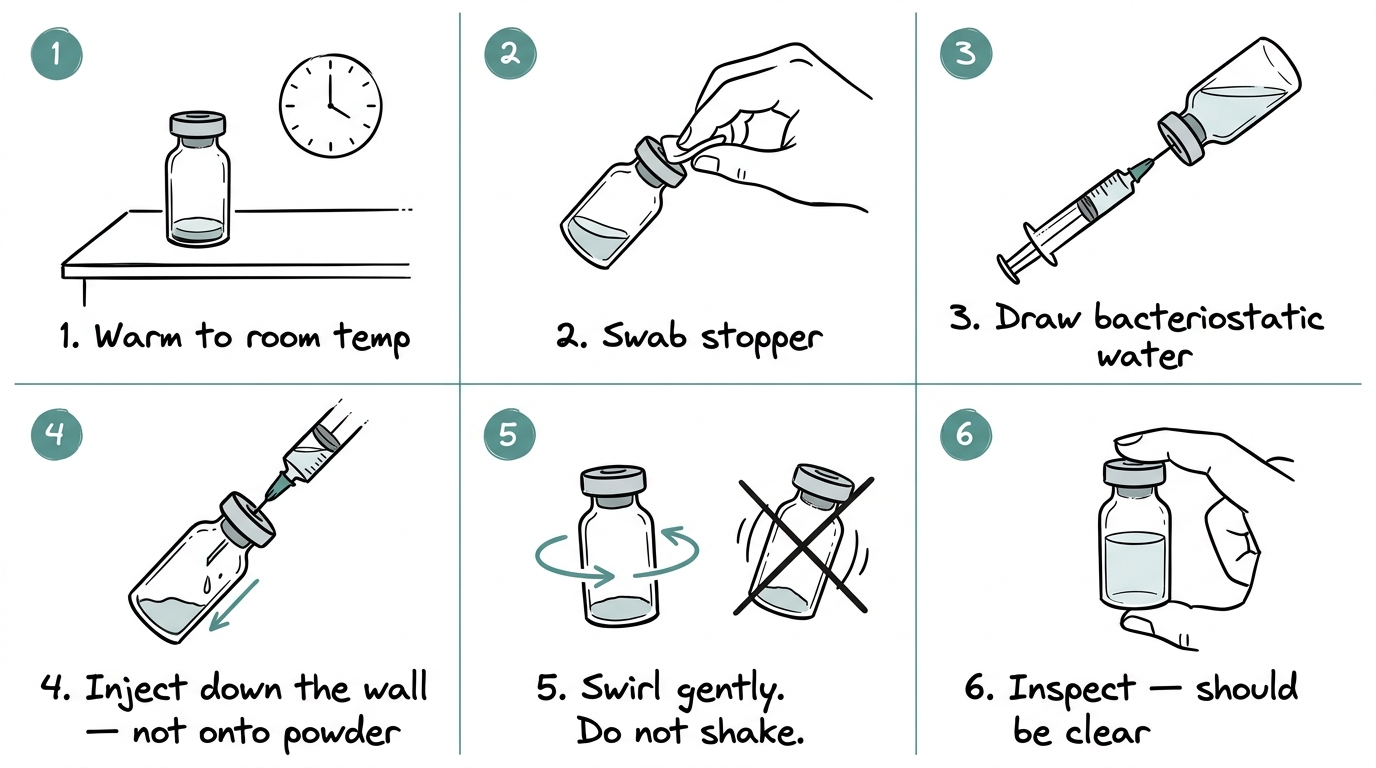
- ●Warm the vial to room temperature.
- ●Swab the vial stopper with an alcohol swab.
- ●Draw the desired volume of bacteriostatic water into a syringe.
- ●Inject the water slowly down the inside wall of the vial — not directly onto the powder.
- ●Swirl the vial gently. Do not shake.
- ●Inspect the solution. It should be clear with no particulates or discoloration.
Two details the diagram cannot show
First: injecting water directly onto the powder can damage the peptide, which is why the down-the-wall rule exists. Second: if the inspected solution is cloudy, shows particulates, or has shifted colour, the peptide has not reconstituted cleanly and should not be used.
The volume of water is a choice the researcher makes. More water means lower concentration, which makes small doses easier to draw accurately. Less water means higher concentration, which makes long-term storage volume smaller.
Dosing math
Two equations, applied in order. Concentration first (peptide mg divided by water mL), then dose volume (target mg divided by concentration). For insulin syringes the standard conversion is 1 mL = 100 units.
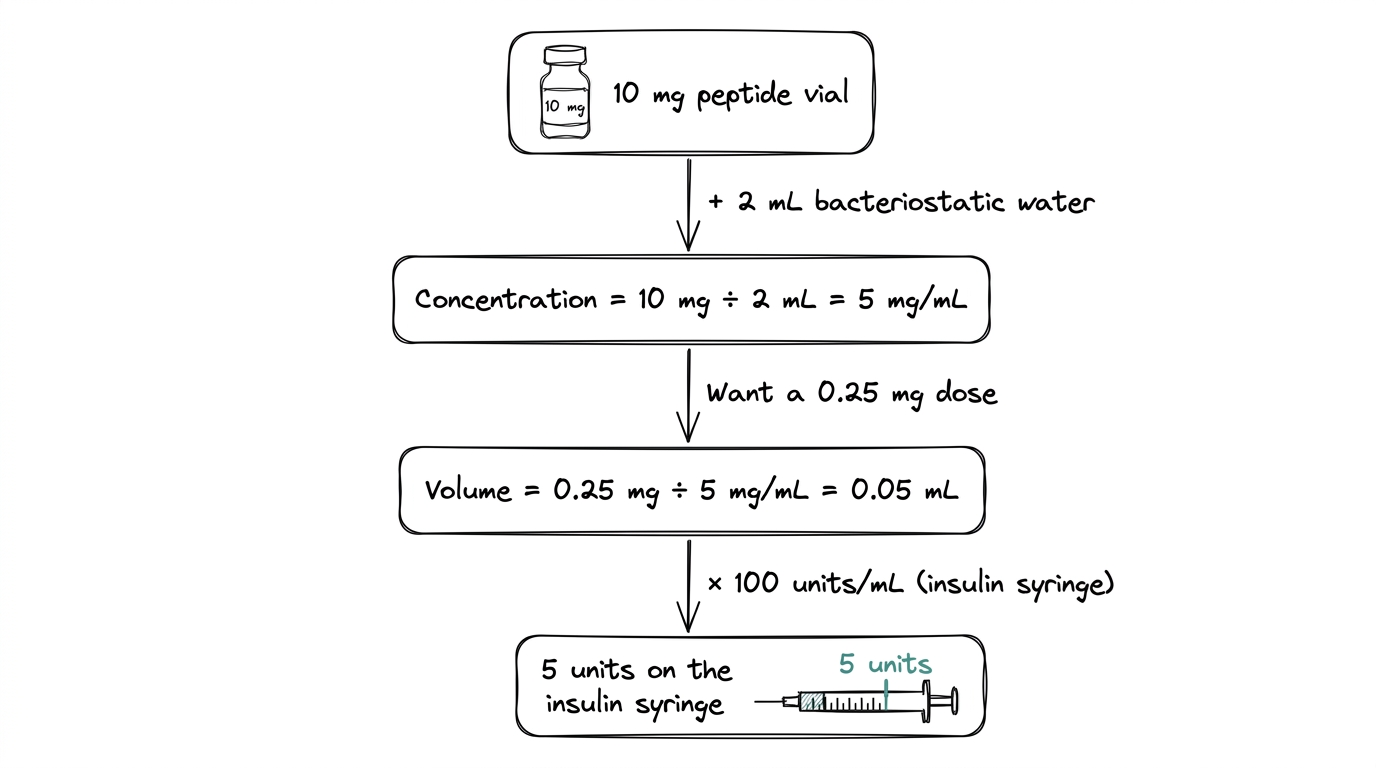
Step 1: Concentration
Peptide amount (mg) divided by water volume (mL) = concentration (mg/mL).
Example: 10 mg vial + 2 mL water = 5 mg/mL.
Step 2: Dose volume
Target dose (mg) divided by concentration (mg/mL) = draw volume (mL).
Example: 0.25 mg divided by 5 mg/mL = 0.05 mL = 5 units on an insulin syringe.
Run this calculation once per vial at reconstitution. Write it down. When doses feel off later, the math is usually the culprit.
Storage
Lyophilized powder and reconstituted solution are two different materials with two different storage windows.
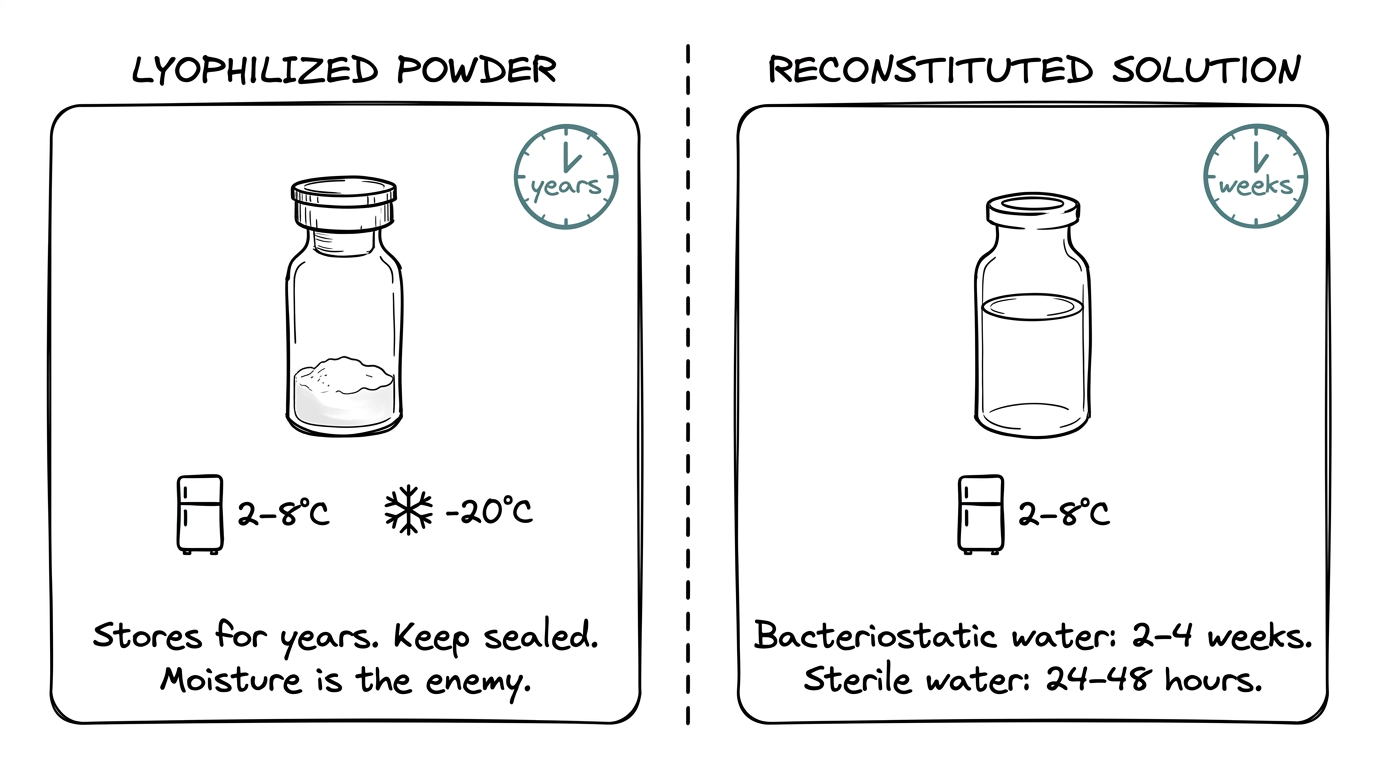
| Category | Lyophilized powder | Reconstituted solution |
|---|---|---|
| Storage temperature | 2-8 degrees C (or colder) | 2-8 degrees C (refrigerated) |
| Shelf life | Years (sealed) | 2-4 weeks (bacteriostatic water) or 24-48 hours (sterile water) |
| Key factor | Keep sealed and dry | Benzyl alcohol preservative in bac water extends life |
The benzyl alcohol in bacteriostatic water is a preservative — that is what buys the multi-week shelf life. Plain sterile water has no preservative, which is why it lasts only 24-48 hours.
Freezing reconstituted solution is possible but introduces freeze-thaw cycle degradation. If a long storage window is needed, store lyophilized and reconstitute in smaller batches.
Shelf life in practice
Three dates per vial. The middle one is the one researchers most often forget to write down.
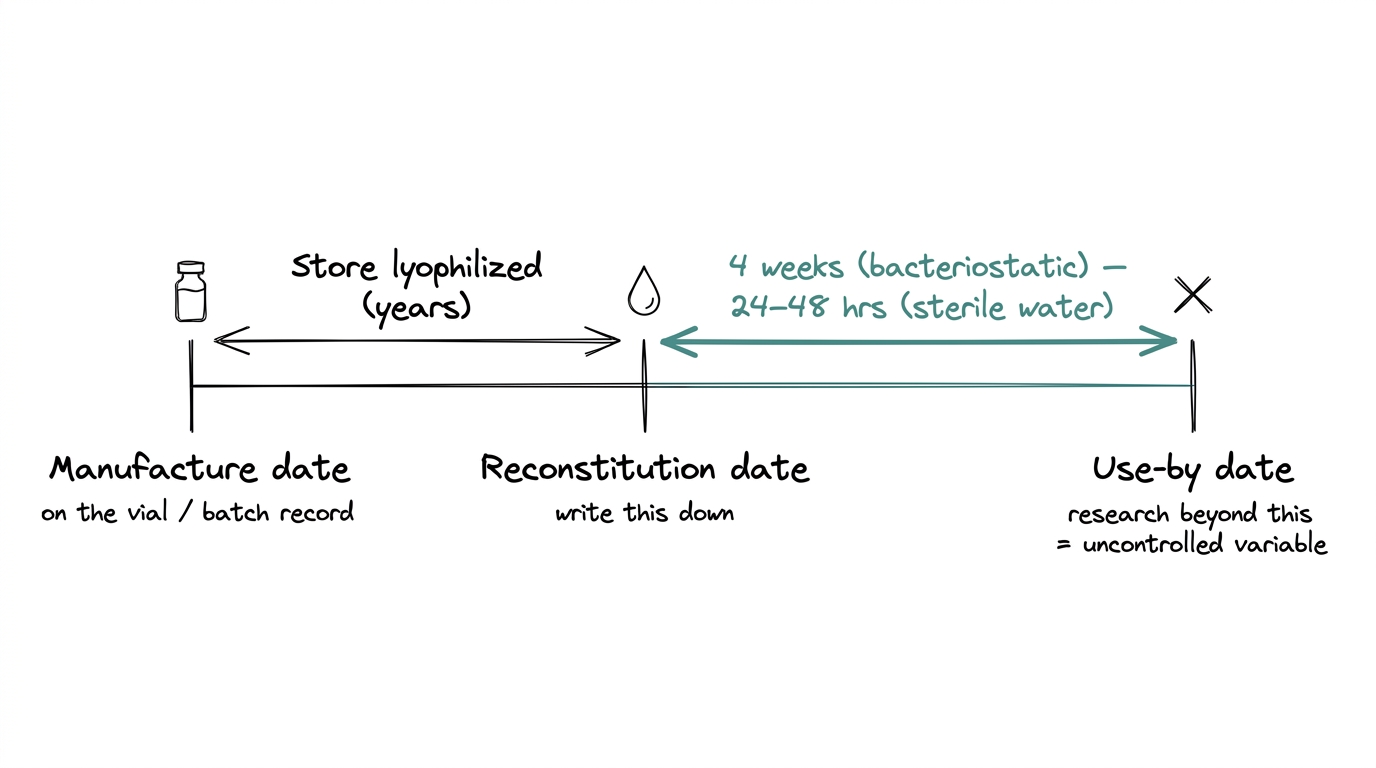
- ●Manufacture date — from the vial or batch record.
- ●Reconstitution date — when you open and reconstitute. Write this on the vial.
- ●Use-by date — four weeks after reconstitution with bacteriostatic water.
A vial that has been sitting reconstituted for six weeks is not the same vial it was at day one. Research using old solution is research with an extra uncontrolled variable.
Sterility without paranoia
Sterile technique comes down to a few habits:
- ●Swab the vial stopper before every draw.
- ●Do not touch the needle or the inside of the syringe barrel.
- ●Use a fresh needle for each draw if possible. At minimum, do not re-use a needle that has been in tissue.
- ●Cap or cover the vial between draws.
These habits keep bacteria out of a solution that will sit in the fridge for weeks. That is the whole goal — anything more is overkill.
Record keeping
A research notebook with four columns beats everything else:
- ●Vial identity (compound, batch, arrival date)
- ●Reconstitution volume and date
- ●Draw volume and date
- ●Observed results
Writing it down is how a researcher catches drift: a dose that was different this week, a solution that looked slightly cloudy, a batch that behaved differently from the last one.
When the compound is the problem
Sometimes the issue is the compound itself. The signs:
- ●The solution does not go clear even after correct reconstitution.
- ●The solution discolors faster than expected.
- ●Results diverge from protocol expectations across multiple vials from the same batch.
- ●Independent testing shows the purity does not match the supplier's claim.
Verify with the supplier before adjusting anything on your end. Good technique cannot compensate for a batch that did not meet spec.
Quick reference
- ●Reconstitute with bacteriostatic water, slowly down the vial wall, no shaking.
- ●Store lyophilized cold and sealed; reconstituted refrigerated and used within 4 weeks.
- ●Run the concentration math once per vial and write it down.
- ●Swab before every draw.
- ●Track vial identity, reconstitution date, and draw log.
- ●These steps solve most handling problems. When they don't, the issue is usually compound quality — check with the supplier.
Frequently asked questions
How do you reconstitute a research peptide?
Warm the vial to room temperature, swab the stopper, draw bacteriostatic water into a syringe, inject slowly down the inside wall of the vial (not onto the powder), swirl gently without shaking, and inspect for a clear solution with no particulates.
What is the difference between bacteriostatic water and sterile water for peptides?
Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, which extends the reconstituted shelf life to 2-4 weeks. Plain sterile water has no preservative and only lasts 24-48 hours after reconstitution.
How long does a reconstituted peptide last?
With bacteriostatic water, reconstituted peptides last 2-4 weeks refrigerated at 2-8 degrees C. With plain sterile water, only 24-48 hours. Lyophilized (powder) peptides stored sealed at 2-8 degrees C or colder can last years.
How do you calculate peptide dosing?
Two steps: first calculate concentration (peptide mg divided by water mL), then calculate dose volume (target dose mg divided by concentration). For insulin syringes, 1 mL equals 100 units. Example: 10 mg vial + 2 mL water = 5 mg/mL; for a 0.25 mg dose, draw 0.05 mL (5 units).
Can you freeze reconstituted peptides?
Freezing is possible but introduces freeze-thaw cycle degradation. If a long storage window is needed, it is better to store the peptide as lyophilized powder and reconstitute in smaller batches as needed.
How do you know if a peptide has gone bad?
Signs include: solution does not go clear after reconstitution, solution discolors faster than expected, results diverge from protocol expectations across multiple vials from the same batch, or independent testing shows purity does not match the supplier claim.
What should you track in a peptide research notebook?
Four columns: vial identity (compound, batch, arrival date), reconstitution volume and date, draw volume and date, and observed results. This log helps catch drift in dosing, solution quality, or batch-to-batch variation.
Next step
Source research-grade peptides with batch documentation
Every vial ships with a batch-specific COA. Pair correct handling with verified compound quality.