Key takeaways
- ●Semaglutide is a GLP-1 receptor agonist that reduces appetite, slows gastric emptying, and regulates glucose — all from a single weekly injection.
- ●It is the most studied and widely available compound in the incretin pathway, serving as the reference baseline for newer multi-receptor agonists.
- ●Research is expanding beyond metabolism into cardiovascular outcomes, inflammation, and dosing optimization.
- ●Effects depend on continued administration — rebound after discontinuation is a known constraint shaping research protocols.
Semaglutide is a synthetic peptide, better known by its brand names Ozempic and Wegovy, that mimics a natural hormone the body releases after eating.
That hormone is called GLP-1. Semaglutide copies its effects and makes them last much longer.
Origin and approval
Novo Nordisk developed semaglutide in the early 2010s as a treatment for type 2 diabetes. The FDA approved it under the brand name Ozempic in December 2017. In June 2021, the FDA approved the same molecule under a second brand name, Wegovy, this time for chronic weight management.
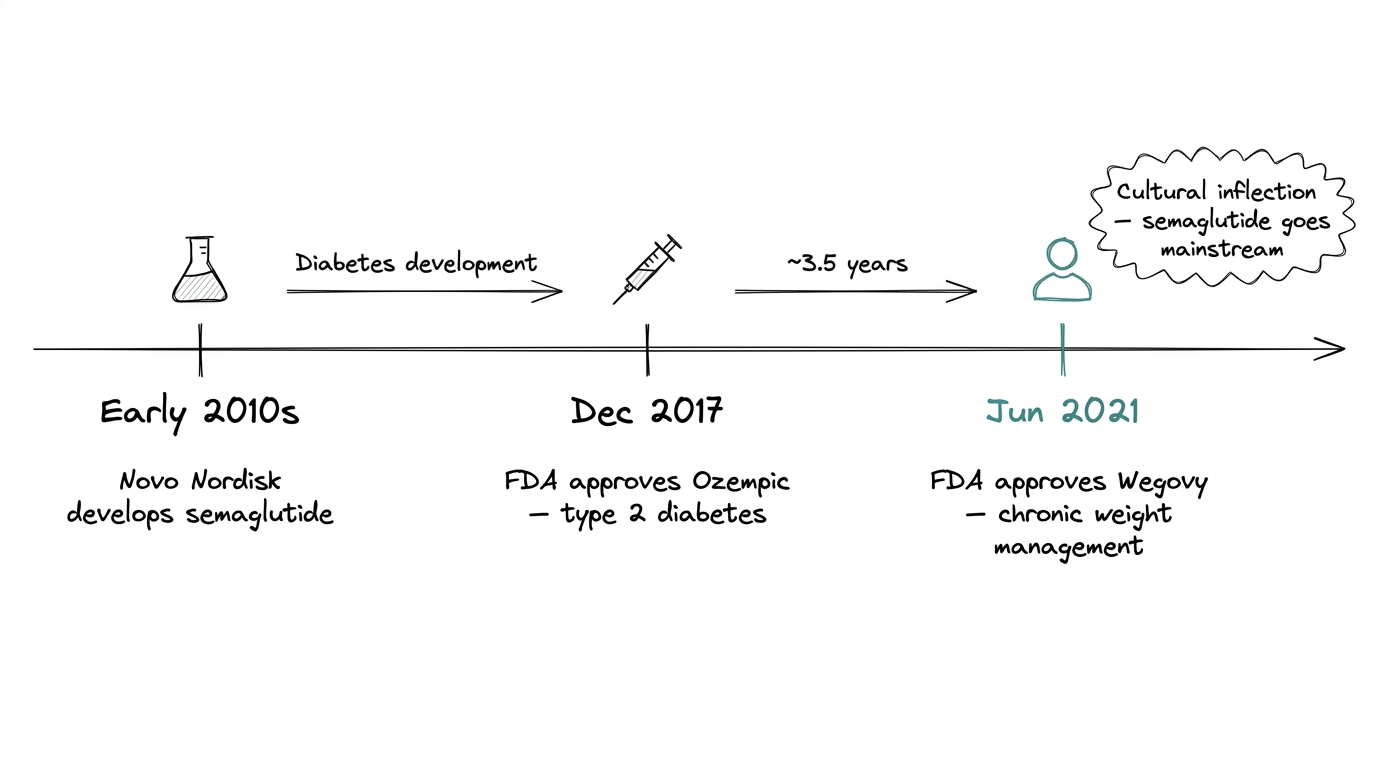
The second approval is what pushed semaglutide into mainstream awareness. The diabetes framing never landed with the general public. The weight-loss one did, fast.
How it works
Semaglutide is a GLP-1 receptor agonist. Two concepts to unpack there.
GLP-1 stands for glucagon-like peptide-1. It is an incretin hormone, meaning the gut releases it after eating, and it tells the rest of the body how to respond to that food.
An agonist is a molecule that activates a receptor the same way the body's own hormone would. So a GLP-1 agonist is a stand-in for GLP-1 that hits the same receptors.
Natural GLP-1 breaks down within minutes. Semaglutide is engineered to resist that breakdown, which is why a single weekly injection can do what the body's own hormone does on a moment-to-moment basis.
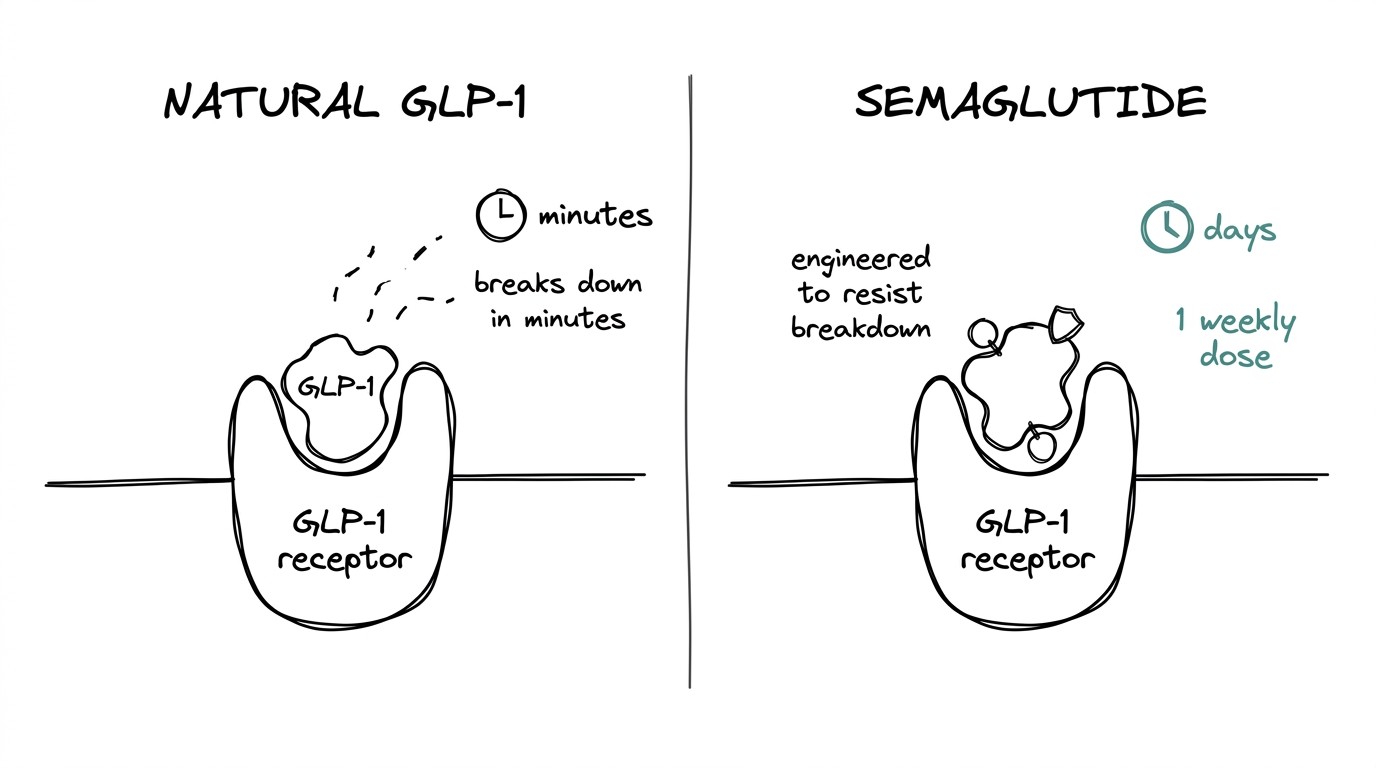
Once active, it drives three mechanisms at once.
Appetite and satiety
Semaglutide activates GLP-1 receptors in the brain, specifically in the hypothalamus, the region that regulates hunger. The result is reduced appetite and a stronger feeling of fullness after eating.
Gastric emptying
It slows how quickly food leaves the stomach. That stretches the window in which the body feels full and reduces hunger between meals.
Glucose regulation
It stimulates insulin release in a glucose-dependent way, meaning it only kicks in when blood sugar is actually elevated, not all the time. It also suppresses glucagon, the hormone that raises blood sugar, which reduces unnecessary blood-sugar spikes.
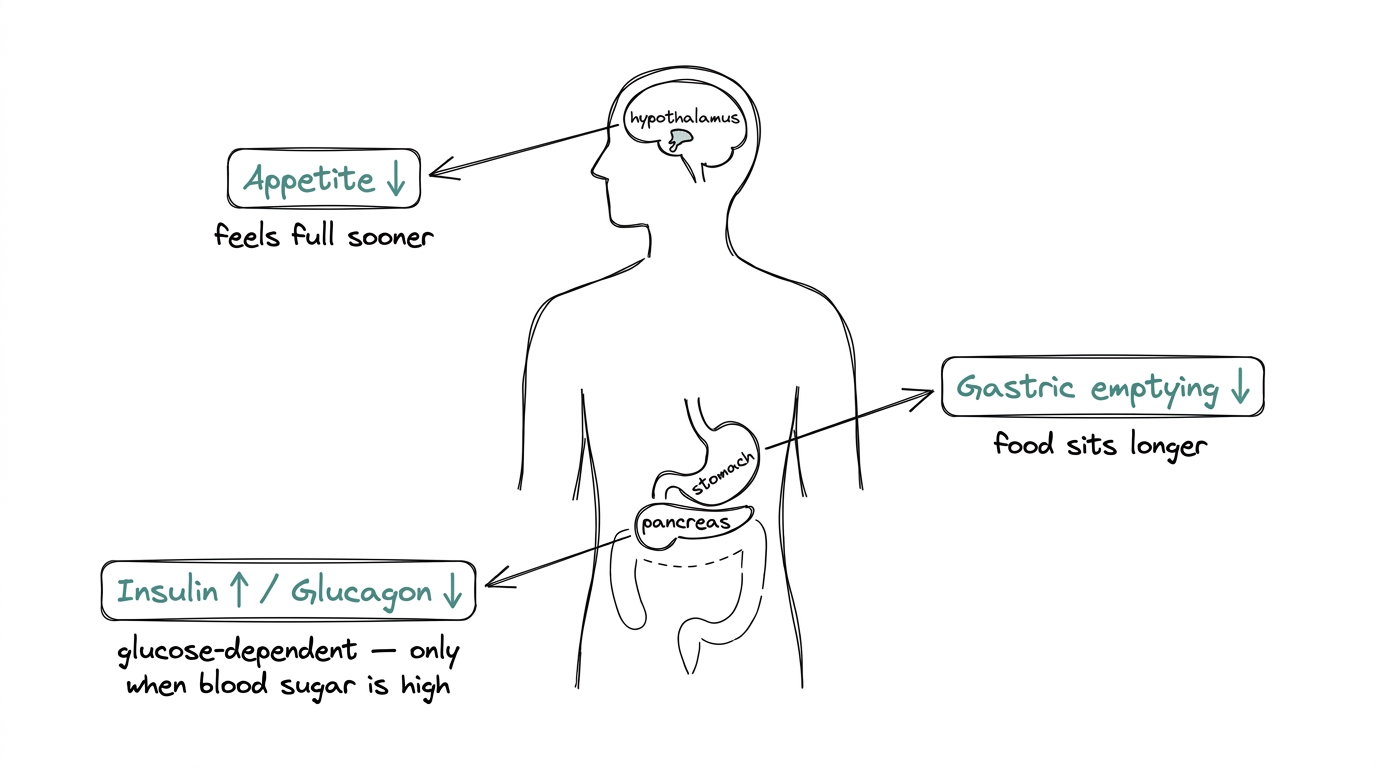
These three effects compound. Smaller meals. Longer satiety. Steadier glucose. That combination is what made semaglutide effective in clinical trials and widely adopted once the weight-management approval landed.
What researchers are exploring
Research interest in semaglutide clusters around four questions.
Durability of effect
How long do the metabolic benefits hold once dosing stops? Published data shows meaningful rebound after discontinuation, and researchers are actively looking at what sustains the change beyond continued administration.
Effects beyond metabolism
GLP-1 receptors show up in tissues well beyond the gut and brain, including cardiovascular tissue, the kidneys, and inflammatory pathways. Studies are examining whether semaglutide influences cardiovascular outcomes and inflammation independently of weight change.
Comparison with next-generation compounds
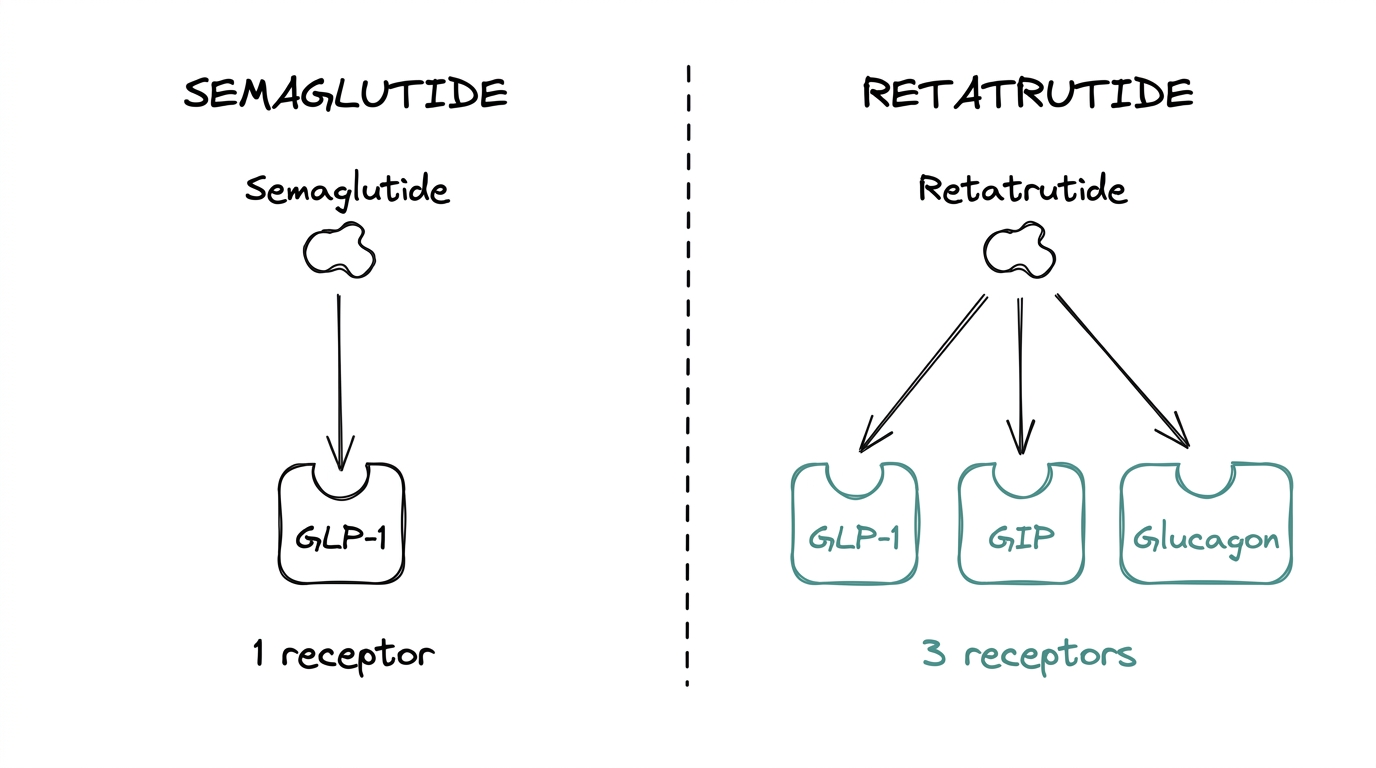
Retatrutide, a triple agonist that adds GIP and glucagon receptor activity on top of the GLP-1 base, is the fastest-growing area of interest in the metabolic peptide category. Semaglutide has become the comparison baseline for every newer compound.
Dosing optimization
Weekly dosing is the current standard. Research is active on titration schedules, break protocols, and combination dosing with other metabolic compounds.
Limits and tradeoffs
Semaglutide has real constraints.
The effect depends on continued administration. When dosing stops, rebound is common, which shapes how research protocols are designed around it.
Gastrointestinal side effects, especially nausea and occasional vomiting or constipation, are the most commonly reported in clinical data. They are dose-dependent and typically ease with slower titration.
Semaglutide also only covers one leg of the incretin pathway. Research interested in GIP or glucagon receptor activity needs a different compound. That is where retatrutide and other multi-receptor agonists come in.
Where to go from here
The GLP-1 reference compound
Semaglutide is the reference compound for GLP-1 research: most studied, most widely available, and the cleanest starting point for anyone new to the incretin pathway.
Newer compounds are more mechanistically complex. More receptors, different questions, different research protocols. Semaglutide is where the learning curve starts.
For a side-by-side look at how it compares to the next wave, see the retatrutide vs ozempic guide.
Frequently asked questions
What is semaglutide?
Semaglutide is a synthetic peptide that mimics the natural hormone GLP-1. It is approved under the brand names Ozempic (for type 2 diabetes) and Wegovy (for chronic weight management).
How does semaglutide work?
It activates GLP-1 receptors to reduce appetite via the hypothalamus, slow gastric emptying, and stimulate glucose-dependent insulin release. Natural GLP-1 breaks down in minutes; semaglutide is engineered to last roughly a week.
What is a GLP-1 receptor agonist?
An agonist activates a receptor the same way the body's natural signal would. A GLP-1 agonist activates GLP-1 receptors — the same ones triggered by the gut hormone released after eating — but with much longer duration.
How does semaglutide compare to retatrutide?
Semaglutide activates one receptor (GLP-1) and achieves roughly 15% weight reduction. Retatrutide activates three receptors (GLP-1, GIP, glucagon) and showed 24-29% weight reduction in trials. Semaglutide is approved; retatrutide is in Phase 3.
What happens when you stop taking semaglutide?
Published data shows meaningful rebound after discontinuation. The metabolic benefits depend on continued administration, which is why durability of effect is an active research question.
What are the main side effects of semaglutide?
Gastrointestinal effects — nausea, occasional vomiting, and constipation — are most commonly reported. They are dose-dependent and typically ease with slower titration.
Next step
Explore GLP-1 research compounds
Research-grade peptides with batch-specific COAs and cold-chain shipping for every order.